The first HIV antibody test became available in 1985. Since then, new HIV antibody tests have been developed and approved by the Food and Drug Administration (FDA). Currently, these antibody tests involve a two-step process utilizing a screening test and, when the screening test is reactive (positive), a confirmatory test.
Those who have put themselves at risk through anal, vaginal, or oral sex, or shared needles, and anyone who has had an occupational exposure may benefit from HIV testing. People may have partners with risk factors, and these people (along with their partners) should consider testing. For occupational exposure, refer to your employer protocol or to Module 5 of this course.
New drug therapies for HIV infection can sustain an infected person’s health for long periods of time. Early detection allows people with HIV the option to receive medical treatment sooner, take better care of their immune system, and stay healthier longer. Additionally, early detection of HIV allows people to take precautions not to infect others.
The Window Period
It is important to remember that HIV antibody testing has a window period. The window period is the time between infection with the virus and when the HIV-infected person develops enough antibodies to be detected by the antibody test. Until the infected person’s immune system makes enough antibodies to be detected, the test will be negative even though the person is infected with HIV.
Unfortunately, there is no way to know how long each infected person will take to develop antibodies. Some infected people are able to produce antibodies as early as 2 weeks after infection and almost everyone will develop enough antibodies to be detected by 12 weeks (3 months) after infection. Therefore, to be sure, people should test 3 months after the last potential HIV exposure.
Because people who are newly infected have so few antibodies to fight HIV, the virus can grow and multiply unchecked. During this time, they can have a large amount of virus in their blood, making them highly infectious for HIV. During the window period it is possible for an infected person to test negative (before they develop antibodies) but still be able to infect another person.
Confidential and Anonymous Testing
HIV testing can only be done with the person’s consent. Consent may be contained within a comprehensive consent for medical treatment. It can be verbal or written, but must be specific to HIV and must be documented. There are some rare exceptions where a person can be tested without their consent.
With confidential HIV testing, the clients give their real name and the information about the testing is maintained in their medical records. The results are confidential. Results and testing information are not released to others except when medically necessary or under special circumstances including when a release is signed for the results to be given to another person or agency. HIV is a reportable condition in most states—including Florida—so confidential HIV results are reported to local public health officials.
Anonymous HIV antibody testing—where clients do not give their name and the person who orders or performs the test maintains no record of the name of the person being tested—is also available in Florida. As part of the informed consent process, a test subject must also be given information on the availability and location of anonymous test sites. Each county health department maintains a list of available anonymous test sites to be disseminated to all people and programs offering HIV testing within their service area (FDOH, 2010).
HIV Antibody Tests
The first HIV antibody test became available in 1985. Since then, new HIV antibody tests have been developed and approved by the Food and Drug Administration (FDA). Currently, these antibody tests involve a two-step process utilizing a screening test and, when the screening test is reactive (positive), a confirmatory test.
Step 1: Screening Test
The first test done on a specimen is a screening test called an enzyme-linked immunosorbent assay test (ELISA, or EIA). This type of test screens for the presence of antibodies to HIV in blood, urine, or oral fluid. Screening tests are inexpensive and highly accurate.
Most HIV antibody screening tests are conventional, in that the specimen is collected from the client and sent to a laboratory for testing. If a screening test is negative (no antibodies detected), the results can be released to the client. If the screening test is reactive (positive) at the laboratory, a confirmatory Western Blot test is conducted on the same sample.
Rapid tests are also screening tests, but they are conducted at the test site, often with the client present, and negative results are available in under an hour. Reactive (antibodies detected) results from a rapid test must be confirmed. This is done because there is a small chance that an HIV screening test may detect proteins related to other autoimmune diseases and react to those proteins with a positive result.
Step 2: Confirmatory Testing
If a rapid test is reactive, an additional specimen must be drawn from the client and sent to the lab for confirmatory Western Blot testing. The HIV Western Blot detects antibodies to the individual proteins that make up HIV. This test is much more specific, and more costly, than the ELISA screening test.
Rapid HIV Test
HIV Rapid Test Kit
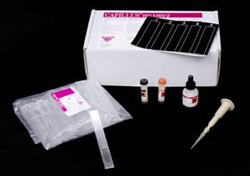
Contents of the CAPILLUS HIV-1/HIV-2 Rapid Test Kit that tests whole blood, serum, or plasma. Source: CDC.
The rapid test is an immunoassay used for screening and it produces quick results, in 20 minutes or less. Rapid tests use blood or oral fluid to look for antibodies to HIV. If an immunoassay (lab test or rapid test) is conducted during the window period (ie, the period after exposure but before the test can find antibodies), the test may not find antibodies and may give a false negative result. All immunoassays that are positive need a followup test to confirm the result (CDC, 2013b). Information about FDA-approved tests and their use in various settings is available through the CDC website.
Home HIV Test
Currently there are only two home HIV tests: OraQuick In-home HIV test and the Home Access HIV-1 Test System. If you buy your HIV home test online make sure it is FDA-approved (CDC, 2013b).
The OraQuick In-Home HIV Test provides rapid results in the home. The testing procedure involves swabbing your mouth for an oral fluid sample and using a kit to test it. Results are available in 20 minutes. If you test positive, you will need a followup test. The manufacturer provides confidential counseling and referral to followup testing sites. Because the level of antibody in oral fluid is lower than it is in blood, blood tests find infection earlier than oral fluid tests. Up to 1 in 12 people may test false negative with oral fluid tests (CDC, 2013b).
The Home HIV Access HIV-1 Test System is a home collection kit that involves pricking your finger to collect a blood sample, sending the sample to a licensed laboratory, then calling in for results a few days later. If the blood tests positive, a followup test is performed right away. This test is anonymous. The manufacturer provides confidential counseling and referral to treatment. The tests conducted on the sample collected at home find infection later than most lab-based tests offered by providers (CDC, 2013b).
A positive test result means you are HIV positive and can infect others who come in contact with your blood, semen, or vaginal fluids. A negative result means there are no antibodies to HIV in your blood at the time of the test. A negative test does not mean you are HIV negative—you may be infected but be in the “window period”—a period lasting up to 6 months in which an infected person has no detectable antibodies in their blood.
Counseling
In most states, HIV test counseling is offered to clients who are at risk for HIV or who request counseling. The goal of HIV counseling is help individuals assess risk, understand test results, and develop a personalized prevention plan.
Florida law requires those who perform HIV tests in county health departments and other registered testing sites obtain the informed consent of the test subject, confirm positive preliminary results with a supplemental test before informing the test subject of the result (except as provided for by statute), and make a reasonable attempt to notify the test subject of the test result. Although Florida law no longer requires pre-test counseling—except in the case of a provider who attends a pregnant woman for conditions related to her pregnancy—the Department of Health recommends that HIV testing be preceded by a pre-test counseling session that includes test purpose and procedures, information about infection and transmission, ramifications of a positive test, reducing risky behavior, available support services, and information on how to obtain test results (FDOH, 2010, 2010b)
Florida law also no longer requires face-to-face post-test counseling; however, it does require that when test results are provided the following be included:
- For positives, information on preventing transmission of HIV, on the availability of appropriate medical and support services, and on the importance of notifying sex and/or needle-sharing partners who may have been exposed. Providers must make a good faith effort to ensure that spouses and former spouses (from the past 10 years) of HIV-infected people are notified that they may have been exposed to HIV infection.
- For negatives, information on preventing the transmission of HIV, if appropriate.
The Department of Health still recommends that face-to-face post-test counseling be provided and that it include: information about the meaning of the test results, potential consequences of a positive result, need for retesting and risk reevaluation, support services, elimination of virus transmission, tuberculosis, and all appropriate referrals.
When a pregnant woman tests positive for HIV, in addition to medical and support services, she should also be referred to the Healthy Start Care Coordination System. For more information on the availability of services, contact the Family Health Line at 800 451-BABY or the Florida AIDS Hotline at 800 FLA-AIDS (FDOH, 2010, 2010b).
