Bob, a 49-year-old construction worker comes to the emergency department with nausea and vomiting that has lasted for two days. He admits to social drinking of 2 beers nightly after work and 4 on weekends for the past 20 years. He states he has been taking 8 tablets of Tylenol daily for the past 2 weeks for pain in his right knee and lower back after a recent fall. He think he may have the flu because he just “doesn’t feel good” and keeps vomiting. Upon physical assessment his skin looks pale yellow and he’s dehydrated. His right knee is swollen and red.
If this patient were your assignment, what labs would you expect to be ordered? What would be your focused physical assessment? What is the primary problem? What is the causative factors? What diagnosis do you suspect?
What Is Acetaminophen?
Acetaminophen is a non-narcotic analgesic and fever reducer. It is available by prescription or over the counter (OTC). Millions of people use over-the-counter (OTC) pain relievers every day to treat minor aches and pains. Usually these medicines are safe and effective, but they can be dangerous and even deadly when they are not taken as directed. That is particularly important with acetaminophen, which is used to relieve pain or reduce fever (FDA, 2017).
Acetaminophen, also called paracetamol, is sold by many names including Tylenol, Panadol, Ofirmev, Acephn, and Mapap. It is used effectively for fever, muscle pain, neck pain, plantar fasciitis, sciatica, and common body aches and pains. It is abbreviated as APAP (for acetyl-para-aminophenol), which is the chemical name, and it is known by 28 brand names in the United States (Mayo, 2017).
It is commonly used in combination drugs for pain as a nonnarcotic analgesic and known as Excedrin, Goody’s Body Pain, Saleto, Exaprin, Levacet, Painaid, Apadaz, Rhinocaps, and Staflex. Acetaminophen is included in OTC medications for acute cold symptoms, respiratory conditions, and headaches. In addition, it is a component of some narcotic prescriptions.
Acetaminophen can be used as a sole medication or as an active ingredient in many OTC and prescription medicines. Acetaminophen is generally safe at recommended doses, but if taken in larger dosages or frequency, it can cause serious and even fatal liver damage. In fact, acetaminophen poisoning is a leading cause of liver failure in this country (Goldberg, 2017).
Acetaminophen Tablets (Generic and Trade)
Acetaminophen is available in both generic and trade-named products. Image Source: Google Images.
Acetaminophen is very effective in relieving pain and fever, which promotes its popularity. It is the generic name of a drug found in hundreds of OTC products including Tylenol, cough and cold medicines, and sleep aids, as well as prescription pain relievers such as Vicodin and Percocet. However, it is not always clear to the consumer that these other named medications contain acetaminophen.
If more than one medication is taken that contains this drug, consumers may inadvertently take more than the recommended dose. Because the risks of acetaminophen-related liver damage are so serious and because the public is often unaware of these risks, the leadership of the National Council for Prescription Drug Programs (NCPDP), the Food and Drug Administration’s (FDA) Safe Use Initiative, and a broad group of stakeholders came together in 2011 to form the Acetaminophen Best Practices Task Group, which produced Version 1.0 of a white paper,* and an update in 2013 (FDA, 2013).
*White paper: an authoritative report or guide helping readers to understand an issue, solve a problem, or make a decision.
The recommendations are intended to make it easier for consumers to (1) identify that their prescription pain reliever contains acetaminophen, (2) compare active ingredients on their prescription and OTC labels, and (3) take action to avoid taking two medicines with acetaminophen. One strategy recommended matching the prescription container labeling with the labeling that already exists for OTC medicines that contain acetaminophen, providing consistency in labeling across all acetaminophen-containing medicines.
The FDA white paper recommends:
- Complete spelling of acetaminophen and all other active ingredients on the pharmacy labels of all acetaminophen-containing prescription medicine and eliminating the use of abbreviations, acronyms, or other shortened versions for active ingredients
- A standardized liver warning label
- A standardized warning label for drugs that may interact with acetaminophen
- Formatting and wording on pharmacy container labels consistent with plain language and health literacy principles
- A stakeholder call to action: “adopt, implement, adhere, communicate and educate” (FDA, 2013)
Acetaminophen, the active ingredient in Tylenol, is also known as paracetamol and N-acetyl-p-aminophenol (APAP). It has been marketed in the United States as an OTC antipyretic (fever reducing) and analgesic (pain reducing) agent since 1953 (Wikipedia, 2018a). Acetaminophen is widely available in a variety of strengths and formulations for children and adults as a single-ingredient product.
Open bottles of Extra Strength Tylenol and Extra Strength Tylenol PM, pain relievers with the active ingredient acetaminophen (paracetamol). Tylenol PM (the white-and-blue tablets) also contains diphenhydramine, a sleep aid. Image Source: Wikimedia Commons.
In the United States acetaminophen is available as 325 milligram (mg) and 500 mg preparations and as a 650 mg extended-release medication intended for arthritis treatment. It comes in many forms, including drops, capsules, and pills, as well as various children’s dissolvable, chewable, and liquid formulations (Medical Dictionary, 2018). It is available in many forms, including solutions, tablets, suppository, powder, capsule, syrup, suspension, elixir, and effervescent tablets.
Acetaminophen is added to many combination prescription narcotic drugs (Vicodin, Percocet) in order to give more pain relief with a minimized dose of the addictive narcotic component. It can also be found in combination with other active ingredients in what are called combination medicines that treat symptoms of colds and flu, allergies, and sleeplessness.
Test Your Knowledge
Acetaminophen is sometimes known as:
- APAP or paracetamol.
- Indomethacin.
- Naproxen.
- Ibuprofen.
Acetaminophen is sometimes an ingredient in combination drugs, such as:
- Celebrex and Enbrel.
- Percocet and Vicodin.
- Lisinopril and benazepril.
- Viagra and Cialis.
Apply Your Knowledge
A mother wants to know what over-the-counter medication she can give her 4-year-old for fever. What are the common brand names for acetaminophen?
Online Resource
Acetaminophen: Side Effects, Drug Interactions, and Natural Anti-Inflammatories (Video 18:11)
https://www.youtube.com/watch?v=a88yGCezgiQ
Answers: a,b
Action of Acetaminophen
How does acetaminophen work to reduce fever and pain?
Acetaminophen is used generally for mild to moderate pain relief and fever reduction. It produces analgesic effects by inhibiting cyclooxygenase (COX) enzymes that catalyze the conversion of arachidonic acid, a fatty acid in cell membranes, to prostaglandins that produce pain, inflammation, and fever.
There are two well-studied isoforms, called COX-1 and COX-2. The isoform COX-1 helps with the synthesis of prostaglandins responsible for protection of the stomach lining, while COX-2 mediates the synthesis of prostaglandins responsible for pain and inflammation. Acetaminophen is a relatively greater selective inhibitor of COX-2 enzymes than COX-1 (Drahl, 2014), similar to Celebrex (celecoxib).
Its antipyretic, fever reducing effects are thought to arise from its actions against COX enzymes in the central nervous system, reducing the amount of prostaglandin E2 in the CNS and thereby lowering the temperature set point in the hypothalamus part of the brain. Its anti-inflammatory properties are much weaker than those of aspirin and other non-steroidal anti-inflammatory drugs (NSAIDs) like ibuprofen, which inhibit both COX-1 and COX-2 and can irritate the stomach and gastrointestinal (GI) tract.
Acetaminophen is thus less effective or appropriate for chronic inflammatory pain conditions such as rheumatoid arthritis, Lupus or Cohn’s; however, it is an excellent choice for osteoarthritis, especially in those patients where aspirin is contraindicated, such as in patients with an aspirin/salicylates allergy or peptic ulcers and other GI sensitivities. Acetaminophen lacks the antithrombotic, blood-thinning properties of aspirin and other NSAIDS, therefore does not inhibit coagulation, an important consideration for pain therapy following minor surgical or dental procedures.
Side Effects of Acetaminophen
All medications have desired action for which the medication is being taken but also have unwanted side effects, and possibly additional adverse reactions (Drugs.com). The desired effects of acetaminophen are fever-reducing and pain relief, but many people may also experience:
- Dark urine
- Itching
- Jaundice
- Loss of appetite
- Nausea
- Upper stomach pain
Online Resources
Acetaminophen Nursing Considerations (7:21)
https://www.youtube.com/watch?v=Kl-7A4XkcPw
Ibuprofen vs. Acetaminophen: What’s the Difference? (3:27)
https://www.youtube.com/watch?v=lIPPILw5dtc
Acetaminophen Oral: Uses, Side Effects, and Interations/Health Tips (1:37)
https://www.youtube.com/watch?v=i9uOqX98HJc
Ibuprofen vs Aleve vs Turmeric vs Tylenol. Pharmacist Compares (Video 11:27)
https://www.youtube.com/watch?v=LB9suniH6MA
Mechanisms of Acute-Overdose Liver Failure
Acetaminophen (APAP, paracetamol) has excellent bioavailability when taken orally and is almost completely absorbed by passive transport in the small intestine. Peak plasma concentrations occur at 30 to 60 minutes with a half-life of approximately 2 hours for therapeutic doses as it becomes distributed fairly evenly throughout the body (except for fat tissue).
Acetaminophen is usually metabolized and inactivated in the liver by adding glucuronidate or sulfate groups to the molecule for increased solubility and excretion via the kidney (Toyoda et al., 2017) (see figure in next section). A small amount is acted on by the cytochrome P450 enzymes, CYP2E1 and CYP1A2, to generate the very reactive and toxic compound, N-acetyl-p-benzoquinone imine (NAPQI) (Pascal, 2017; Yang, 2017).
Under normal conditions, the liver will detoxify the NAPQI intermediate by adding a glutathione group with the enzyme glutathione-S-transferase (GST), leading to subsequent excretion through the kidney (see figure). A safety warning for people who have chronic and elevated alcohol use, those who take isoniazid for tuberculosis, or those who take St. John’s wort, acetaminophen may induce elevated levels of the CYP2E1 enzymes and generate relatively more of the toxic NAPQI compound, depleting the liver’s store of protective glutathione antioxidant molecules (Wikipedia, 2018b).
This can result in a higher level of reactive oxygen and eventual necrosis in liver cells (Jaeschke et al., 2012). Likewise, in cases of overdose, intentional or not, the levels of acetaminophen can increase for up to 4 hours, and the NAPQI produced can saturate the GSH detoxification pathway and spill over to cause liver and renal toxicity. The NAPQI compound is toxic because it chemically reacts with other proteins and DNA in the cell, destroying their activity and eventually the mitochondrial function that generates ATP energy (Jaeschke, 2012).
Acute liver toxicity may exhibit clinical symptoms of severe stomach pains, nausea, vomiting, and upper-right-quadrant abdominal sensitivity, leading some to naively take more acetaminophen for pain relief. Liver failure may happen several days after an overdose, and unfortunately the patient may assume the overdose has been resolved, leaving only immediate liver transplant as a saving option.
Online Resources
FDA Acetaminophen doses over 325 mg might lead to liver damage (Video 4:22)
https://www.youtube.com/watch?v=iB-k01s-v2U
Does Acetaminophen Cause Liver Damage? (3:37)
https://www.youtube.com/watch?v=K7WCSwEWjdk
Paracetamol/Acetaminophen Metabolism
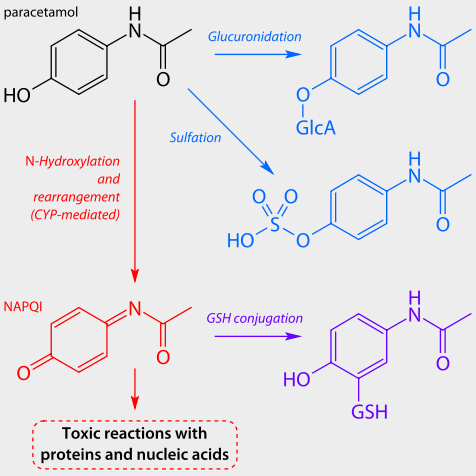
Image Source: Wikimedia Commons.
Our patient Bob has remained in the ED for testing, and results show the following: Vital signs: T, 99.4F; BP, 142/88; R, 20; P, 98.
Physical Exam: sclera mild icterus/yellow
Labs:
Liver enzymes ALT 558
AST 525
bilirubin 41
glucose
blood acetaminophen 58 µg/ml
Normal
4–51 IU/L
15–45
< 17 µmol/L
50
0
The admitting diagnosis is acetaminophen toxicity and Bob is admitted to the hospital. He is given an IV D5W for hydration and given N-acetylcysteine (NAC) as the antidote for acetaminophen overdose.
What put our patient at risk for this? Due to his fall and resultant knee pain he had been self-medicating with a higher than normal dose of acetaminophen. In addition, due to his high alcohol intake, the liver was not able to metabolize the high doses of acetaminophen and created hepatotoxicity.
Patient education and stabilization will be done before he can be discharged. His knee will also require a workup, evaluation, treatment, and appropriate pain management.
Recommended Dosage
Due to data that revealed unintentional overdoses of acetaminophen, in 2011 the U.S. Food and Drug Administration (FDA) asked drug makers to limit the amount of acetaminophen in prescription combination pain relievers. The goal is to reduce accidental overdoses and the severe liver injury that can follow. This request is hoped to limit the amount of acetaminophen in these products to 325 mg per tablet, capsule, or other dosage unit, making these products safer for patients (FDA, 2014).
A Boxed Warning highlighting the potential for severe liver injury and a Warning highlighting the potential for allergic reactions (eg, swelling of the face, mouth, and throat; difficulty breathing; itching or rash) are being added to the label of all prescription drug products that contain acetaminophen. Manufacturers must also update the labels of all prescription products containing acetaminophen with a Boxed Warning on the risk of severe liver injury if too much acetaminophen is taken or consumed with alcohol (FDA, 2011).
The new dose restriction does not apply to over-the-counter (OTC) pain relievers and cold medicines that contain acetaminophen. Normally, the maximum level allowed for these products is 500 mg, although a few extended-action pain relievers that are taken less frequently can go up to 650 mg (FDA, 2011).
The dosage requirement affects dozens of prescription analgesics that contain both acetaminophen and another ingredient, typically opioids such as codeine, oxycodone, and hydrocodone. Some of these combination products now have as much as 750 mg of acetaminophen per dose.
Patients prescribed analgesics with acetaminophen at doses above 325 mg can safely continue to take them under a physician’s supervision. The key to safety is not exceeding the maximum daily dose of 4000 mg, whether it comes in the form of prescription medications, OTC medications, or both (FDA, 2017).
Although the FDA is considering decreasing the maximum recommended dose of acetaminophen, the currently recommended dose for adults and children over 12 years of age is 650 mg to 1000 mg every 4 to 6 hours as needed, but not to exceed 4000 mg in a 24-hour period. The recommended dose of extra-strength acetaminophen (which contains 500 mg per tablet) is 1000 mg every 4 to 6 hours (Medical Dictionary, 2018).
Did You Know . . .
Two extra- strength acetaminophen tablets taken more than 4 times a day will produce an overdose, and it only takes a few days of exceeding the maximum dose to cause at least some liver damage.
If the patient also consumes alcohol, the risk of damage increases further. Because of this increased risk, the maximum dose of acetaminophen for a person who consumes more than two alcoholic drinks a day should be decreased to 2 grams per day (Medical Dictionary, 2018).
Test Your Knowledge
The current recommended maximum 24-hour dose of Tylenol for adults is:
- 1000 mg.
- 2000 mg.
- 4000 mg.
- 6000 mg.
People who consume more than two alcoholic drinks per day should:
- Consider rehab before taking any pain medications.
- Take no more than 2000 mg per day.
- Take only NSAIDs for pain.
- Not take acetaminophen at all.
Answer: c,b
Pediatric doses are based on age and weight, usually 10 to 15 mg/kg of body weight for a single dose and no more than five doses in a 24-hour period (Ji et al., 2012). FDA approval for intravenous acetaminophen use in infants and children is also now available (FDA, 2013). The maximum recommended dose of extended-release acetaminophen is 1300 mg every 8 hours. Acetaminophen extended-release, extra-strength, and arthritis formulas are not recommended for children less than 12 years of age.
In 2011 an FDA advisory panel announced that pediatric doses of acetaminophen should be labeled “fever reduction only” in children under age 2. The panel found there was too little evidence to label OTC acetaminophen for pain relief in infants under age 2, although doctors often prescribe the drug for this purpose. The advisory panel also re-emphasized that prescribing acetaminophen for children should be based first on weight and then on age.
The panel also advised the FDA to require that:
- Bottles of infant acetaminophen carry dosing instructions for ages 6 months to 2 years
- Bottles of liquid acetaminophen be changed to make it harder for kids to open and take an accidental overdose
- Acetaminophen bottles to come with a liquid measuring device clearly marked in milliliters
The FDA announced that an additional less-concentrated form of liquid acetaminophen marketed for “infants” (160 mg/5 mL) would be available at local stores, packaged with an oral syringe rather than a dropper. Until then, liquid acetaminophen marketed for infants had only been available in higher concentrations (80 mg/0.8 mL or 80 mg/1 mL). This change in the concentration affects the amount of liquid given to an infant and should be especially noted if someone is accustomed to using the more concentrated form (Drugs.com, 2018).
The pharmacy industry also announced it would put flow restrictors on liquid acetaminophen bottles to prevent unsupervised children from drinking large amounts of the medication. Companies will provide clearly marked syringes with all products for kids ages 3 and younger and will add clearly marked dosing cups to all products for kids ages 2 to 12.
The maximum amount of acetaminophen in a prescription tablet, capsule, or other dosage unit will be limited to 325 mg. However, the total number of tablets or capsules that may be prescribed and the time intervals at which they may be prescribed will not change as a result of the lower amount of acetaminophen. For example, for a product that previously contained 500 mg of acetaminophen with an opioid and was prescribed as 1 to 2 tablets every 4 to 6 hours, will now contain 325 mg of acetaminophen and the dosing instructions can remain unchanged (FDA, 2017).
It is also advised that if a patient takes the maximum dosage of acetaminophen allowed for two weeks, and the problem isn’t resolved, it is time to review with the physician the cause and better treatment for the pain. Higher doses such as 500 mg/tablet have also not show to deliver better pain reduction than 325 mg/tablets. Unfortunately, for non-prescription products, the “extra-strength” label that delivers 500 mg/tablet is still available (Consumer Reports, 2014).
Key Points
- Advise patients not to exceed the acetaminophen maximum total daily dose of 4 grams/day.
- Severe liver injury, including cases of acute liver failure resulting in liver transplant and death, has been reported with the use of acetaminophen.
- Educate patients about the importance of reading all prescription and OTC labels to ensure they are not taking multiple acetaminophen-containing products.
- Advise patients not to drink alcohol while taking acetaminophen-containing medications.
- Rare cases of anaphylaxis and other hypersensitivity reactions have occurred with the use of acetaminophen.
- Advise patients to seek medical help immediately if they have taken more acetaminophen than directed or experience swelling of the face, mouth, and throat, difficulty breathing, itching, and rash.
Source: FDA, 2017.
For a list of marketed acetaminophen-containing prescription products affected by the new dosage unit limits, see Drugs.com (2018).
Acetaminophen Frequency of Use
Acetaminophen is the most widely used analgesic medication in the United States despite its potential for hepatotoxicity. Over the past few years, the use of combination prescription products has increased dramatically, and is the biggest cause of acetaminophen overdose (Hodgman & Garrard, 2012).
It is estimated American consumers purchased more than 28 billion doses of products containing acetaminophen, of which:
- Single-ingredient OTC products (eg, Tylenol) represented 8 billion doses.
- Combination OTC products (eg, NyQuil, Theraflu), represented more than 9.7 billion doses.
- Acetaminophen-containing prescription narcotics represented 11 billion doses.
Almost 80,000 people in the United States are treated in Emergency Departments annually due to accidental overdose of acetaminophen (Consumer Reports, 2014). Over 600 OTC and prescription medication products contain acetaminophen.
In the last decade use of these combination prescription products increased 38%. There were more than 182 million prescriptions for combination prescription products and the most frequently used acetaminophen-containing prescription product is Vicodin (hydrocodone/acetaminophen combination). It has been the most frequently dispensed prescription drug since 1997 (FDA, 2011).
The U.S. Centers for Disease Control estimate that more than 980 deaths occur each year due to acetaminophen-containing combination drugs and at least 300 people die as a direct result from acetaminophen-only poisoning (Miller, 2013). Since 2006 these death numbers represent more fatalities due to acetaminophen than all other OTC pain relievers and surpassed those who died from intentional overdose to commit suicide.
Online Resource
Acetaminophen Toxicity (Video 6:09)
https://www.youtube.com/watch?v=xc2QAS_kF0A
Apply Your Knowledge
What are the risk factors that can augment the potency of acetaminophen and cause liver damage?
