*This course has been retired. There is no replacement course at this time. Please click here to view the current ATrain course listings.
Atherosclerosis is the disease behind the disease. When atherosclerotic processes take hold in the arteries that supply blood to the heart, the condition becomes coronary artery disease (CAD). Atherosclerosis is a degenerative disorder that injures the inner walls of large arteries. In atherosclerosis, thick abnormal patches called plaques accumulate at scattered locations along the artery’s innermost endothelial layer. The plaques are disorganized masses filled with cholesterol, lipids, and other cells, all covered by a white fibrous coating (Mitchell & Schoen, 2009).
Atherosclerotic plaques narrow an artery and hinder blood flow. The result is that the surface of a bulging plaque sometimes tears, exposing material that stimulates clot formation. Clots and ruptured plaque material can then break away from the wall, be carried by the blood, and clog arteries downstream.
Atherosclerosis damages tissues throughout the body:
- Blockages in the carotid arteries can reduce or block blood flow to the brain, causing a stroke.
- Obstructions in the peripheral arteries in the legs can cause claudication (pain when walking), gangrene, and deep vein thrombosis (DVT).
- Resultant DVTs can travel to the lungs, causing a pulmonary embolism and respiratory arrest.
- Atherosclerotic obstructions in the intestines cause ischemic bowel.
- In the coronary arteries, atherosclerotic obstructions cause heart attack (NHLBI, 2016c).
Causes of Atherosclerosis
Causes of atherosclerosis include fatty streaks, foam cells, atherosclerotic risk factors, and plaque destabilizers.
Fatty Streaks
Beginning in childhood, a cascade of events slowly and quietly leads to the development of atherosclerosis. Children develop fatty streaks along the walls of their large arteries largely from diets high in fat. These streaks are sites where lipoprotein particles are protected from direct contact with the blood. The lipoproteins become oxidized into destructive molecules and the resulting oxidants injure nearby cells.
Foam Cells: Overwhelmed Macrophages
In the arterial walls, leukocytes (white blood cells) are attracted to the areas of cell injury, and the incoming leukocytes initiate a local inflammatory reaction. Some of the attracted leukocytes are macrophages. Macrophages are clean-up cells, and they begin to engulf the local lipids. When macrophages are overwhelmed by the lipids in their vicinity, they become bloated with fatty debris. The accumulation of fat-filled macrophages, which are called foam cells, is a characteristic of atherosclerotic plaque.
In a person with a healthy balance of blood fats such as low blood levels of LDL cholesterol and high blood levels of HDL cholesterol, there is only a modest accumulation of lipids in arterial walls. The available macrophages can swallow and cart off sufficient lipids from the fatty streaks to avoid a lipid buildup, and few foam cells accumulate. When this and the other lipid-removal mechanisms are working smoothly, the amount of sequestered lipid can be controlled and atherosclerosis is minimal. Unfortunately the American diet is high in fat and generally overwhelms the macrophages’ ability to clean up damaged arteries.
A total lipid profile for Mr. Hansen revealed total cholesterol >200 mg/dL and low HDLs <25 mg/dL. Atherosclerotic plaques had built up over time and left only a 1% space for blood flow at the junction of the LAD.
Test Your Knowledge
Atherosclerotic plaques are filled mainly with:
- Blood clots.
- Lipids and other cells.
- Embryonic arterial wall tissue.
- Glycogen.
Foam cells are:
- Macrophages overfilled with lipids.
- Lipid droplets.
- Vacuolated red blood cells.
- Necrotic, “foamy-looking” smooth muscle cells.
Online Resource
Atherosclerosis [13:44]
https://www.youtube.com/watch?v=g3kDdg8r6NY
Answers: B,A
Atherosclerotic Risk Factors
A variety of conditions can make the lipid removal systems inefficient; they are known as atherosclerotic risk factors. Atherosclerotic risk factors include smoking, diabetes, hypertension, obesity, physical inactivity, diet high in saturated fats, stress, and the accumulated wear of old age. When these conditions interfere with lipid removal, atherosclerosis can slowly clog arteries (Libby, 2008).
In atherosclerosis, the body’s lipid removal systems are working poorly. Foam cells die before they can remove lipids, and a core of necrotic cells forms inside the expanding yellow streak. The body attempts to repair the damage through collage, which enter the mass and begins to create a meshwork with other extracellular matrix materials. By this point, the yellow streak has become an atherosclerotic plaque.
Normal and Atherosclerotic Arteries
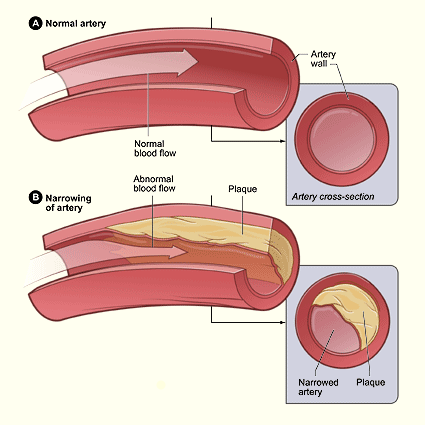
(A) Normal artery with normal blood flow; (B) artery with plaque buildup and diminished blood flow. Source: NHLBI, 2016b.
Plaque Destabilizers
Gradually, atherosclerotic plaques thicken. Internally, the plaques are disorganized and weak. As part of the local repair efforts, a network of small blood vessels grows into the enlarging plaque. The new vessels tend to be leaky due to the inflammatory process, which dilates blood vessels to more easily allow passage of white blood cells, macrophages, and signaling chemicals. The result is that hematomas and clots form inside the plaque. Some regions in the plaque end up with little oxygen and few nutrients. In these poorly oxygenated areas, the local cells—mainly leukocytes and smooth muscle cells—die and create additional necrotic pockets. As in many tissues with chronic inflammation, calcium salts are slowly deposited, making parts of the plaque brittle, causing arteriosclerosis, or hardening of the arteries.
Along with hematomas, these necrotic pockets make the entire plaque susceptible to erosion, crumbling, and rupture. Tears in the fibrous coat of a plaque allow the extracellular matrix inside to come in contact with blood. This stimulates blood-clotting mechanisms and clots form along the edges of the eroded plaque.
Many people go through their entire lives unaware that they have atherosclerosis; however, even asymptomatic atherosclerosis is a constant threat because plaques continue to enlarge. One consequence of this continued growth is that the pressure of an enlarging plaque weakens the muscular layer of the arterial wall and makes the region susceptible to ballooning into an aneurysm (rupture of the wall).
Another consequence is that atherosclerotic plaques become less stable. At some point, the pressure of local blood flow can push the clots and plaque debris downstream to become artery-clogging emboli. The obstructed arteries leave their surrounding tissues ischemic, and the ischemic tissue eventually dies (Libby, 2008).
Test Your Knowledge
As they age and thicken, some atherosclerotic plaques accumulate deposits of:
- Copper.
- Potassium.
- Sodium.
- Calcium.
Answer: D
Atherosclerotic Plaque
Atherosclerotic plaques are white or yellowish bulges filled with cells, cholesterol, and cellular debris. When the surface of a plaque becomes abraded or torn, clots form, and these areas appear red-brown. Individual atherosclerotic plaques are 0.3 to 1.5 cm in diameter, but adjacent plaques can merge into larger lesions.
Atherosclerotic plaques develop in patches, and they are more likely to develop in places where epithelial cells have been injured. Regions of blood turbulence, such as arterial branch points, have chronically injured epithelial cells, and, in atherosclerosis, plaque tends to develop near branch points inside large arteries.
Certain arteries are more likely to have atherosclerotic plaques than others. Here, in order, are the arteries most likely to have significant atherosclerosis:
- Abdominal aorta
- Coronary arteries
- Popliteal arteries (behind the knees)
- Internal carotid arteries
- Arteries in the circle of Willis, which supply blood to the cerebral hemispheres of the brain (Mitchell & Schoen, 2009)
Atherosclerosis of the Coronary Arteries
The two main areas most commonly thickened by atherosclerosis are the lower abdominal aorta and the coronary arteries. Within the coronary arteries, the branch points, shaped like forks in the road, tend to accumulate the thickest buildup of atherosclerotic plaque.
Atherosclerotic Artery with Narrowed Lumen
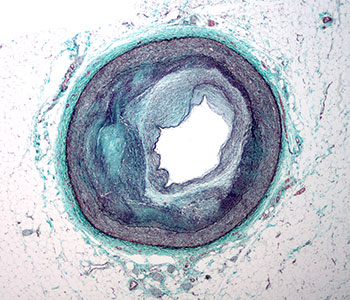
Micrograph of the distal right coronary artery with complex atherosclerosis and luminal narrowing. Source: Wikimedia Commons.
Atherosclerotic plaque forms as a mass inside the arterial wall. Initially, this mass causes a bulge on the outside of a coronary artery, increasing the artery’s overall diameter. Later, as the disease progresses, the plaque begins to bulge into the lumen (inner passageway) of the artery, and at this stage, the plaque reduces the effective inner diameter of the artery and the stenosis can be detected by angiography.
Coronary arteries that have lost 50% to 75% of their inner diameter can no longer deliver sufficient blood flow to meet the increased needs of heart muscle during exercise. That level of stenosis will lead to ischemic symptoms, typically angina, when the patient exercises. If atherosclerotic plaque thickens sufficiently to fill 80% to 90% of the diameter of a coronary artery, ischemic heart symptoms appear even when the person is resting (Antman et al., 2008).
Over time, the section of blood vessel that lies beyond a narrowed segment will dilate, any small collateral arteries will widen, and new collaterals will grow in efforts to deliver life-giving oxygen. In these ways, the arteries of the heart can partly compensate for a narrowed coronary artery when the stenosis has developed gradually.
As brilliant as the body is in adapting for survival, there is no natural protection against sudden arterial narrowing, such as the obstruction produced by clots or debris from a ruptured plaque. Sudden narrowing can lead to ischemia and heart muscle death. This is the most common cause of the death of heart muscle: greater than 95% of all MIs are caused by blood clots and debris from atherosclerotic plaque.
Test Your Knowledge
Atherosclerotic plaques are likely to accumulate:
- At branch points and forks inside large arteries.
- In capillaries and small arterioles.
- On the surface of mucous membranes.
- In the large veins, such as the vena cava.
The arteries that supply blood to the heart can partly compensate for a narrowed artery when the stenosis:
- Is caused by a sudden blood clot.
- Is caused by a sudden disruption of atherosclerotic plaque.
- Has been caused by a sudden vasospasm.
- Has developed gradually.
Heart muscle cells that become ischemic will:
- Die within 1 to 2 minutes unless they are immediately reperfused.
- Stop working but can “hibernate” for weeks and then revive when reperfused.
- Die in 20 minutes to 4 hours if not reperfused.
- Keep working for 2 to 5 days before they slowly die.
Answers: A,D,C
Effects of Atherosclerosis on the Heart
Normally, when heart muscle works harder, the blood vessels inside the heart—those arteries downstream from the main coronary arteries—dilate to increase blood flow to the muscle cells. However, if atherosclerosis has narrowed the coronary arteries, dilation of the downstream arteries is not enough to prevent ischemia (Antman et al., 2008).
The region of heart muscle that becomes ischemic depends on which artery has been blocked. Approximately half of all MIs are caused by blockages of the left anterior descending artery, 30% to 45% are caused by blockages of the right coronary artery, and 15% to 20% are caused by blockages of the left circumflex artery (Burke & Virman, 2008). After a sudden blockage, the heart muscle cells fed by these arteries become ischemic and, unless they are quickly reperfused, ischemic heart muscle cells will die in 20 minutes to 4 hours, depending on the existing collateral circulation.
Myocardial Infarct in the Wall of the Left Ventricle
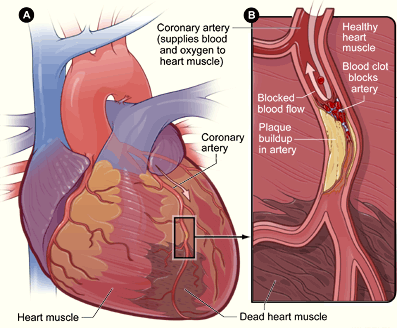
(A) Blood flowing down the left anterior descending (LAD) branch of the left coronary artery has been obstructed. Heart muscle beyond the blockage has died from prolonged ischemia (brown patch). (B) A cutaway diagram of the LAD shows plaque (yellow) that had gradually narrowed the arterial lumen. Later, a blood clot, broken loose from upstream plaque, has become wedged into the narrowed section of the LAD, completely obstructing blood flow Source: NIH, 2016.
Symptoms of CAD
Coronary artery disease can become a chronic disease called stable angina. It can also give rise to sudden cardiac emergencies called acute coronary syndromes. Acute coronary syndromes range from temporary episodes of significant ischemia (unstable angina) to permanent heart muscle damage (myocardial infarction) to sudden cardiac death.
Stable Angina
In stable angina, coronary arteries are partly occluded. At rest, dilation of the downstream arteries allows sufficient blood flow to meet the demands of the heart muscle cells. However, during exercise, the increased oxygen and nutrients needed by the heart exceed the capacity of the already-dilated arteries. Therefore, when the person exercises, the heart muscles become ischemic, and typically the person feels angina. In most cases, the ischemia of stable angina does not cause significant muscle cell death.
Symptoms in stable angina occur when the demands on the heart exceed the blood flow through pre-existing stenosis. These symptoms occur predictably, whenever the patient’s heart accelerates to a certain level of activity. In stable angina, the event that initiates symptoms is generally external such as exercise, stress, or cold weather. Internally stressors such as psychological stress can also create hypertension and resultant demands on ischemic heart vessels.
Acute Coronary Syndromes
In acute coronary syndromes, the event that initiates symptoms is an internal change in the atherosclerotic plaque in the patient’s coronary arteries. When a patient suffers an acute coronary syndrome:
- Plaque has expanded, eroded, or ruptured, or
- A thrombus has moved or broken loose, or
- A coronary artery has suddenly tightened in a vasospasm.
When arterial obstruction persists, causing sufficient ischemia to kill muscle cells, this is called a myocardial infarction (MI, or heart attack). After an MI, intracellular proteins leak from the damaged cells and circulate in the bloodstream. An MI can be diagnosed by finding cardiac-specific intracellular proteins (cardiac biomarkers) in the blood of a person who has the signs and symptoms of an acute coronary syndrome.
Myocardial infarctions can be definitively diagnosed in symptomatic patients from blood tests. When heart muscle cells die, heart-specific intracellular molecules (cardiac biomarkers such as troponin) leak into the bloodstream.
Sudden Cardiac Death
The abrupt release of atherosclerotic thrombi that causes myocardial ischemia can also trigger fatal ventricular arrhythmias. This appears to be the critical event behind most cases of sudden cardiac death, a condition in which patients die unexpectedly and within minutes of the onset of symptoms (Schoen & Mitchell, 2009). This fatal symptom is the end stage of CAD and can only be altered with cardiopulmonary resuscitation.
Mr. Hansen had developed stable angina over years of sedentary lifestyle, high-fat diet, and work stress. Unstable angina had developed quickly as new stress worsened at work and a new baby was born to the busy family. Even happy events such as marriage or a baby can result in stress on the body.
Test Your Knowledge
The chest discomfort of stable angina is often described as:
- Sharp pain.
- Fleeting pain.
- Squeezing or tightness.
- Made worse by the movements of coughing or breathing.
Cardiac biomarkers are intracellular heart muscle molecules that:
- Are released into the blood when heart muscle cells die.
- Are released into the blood all the time.
- Become dense and visible in CT scans when heart muscle cells first becomes ischemic.
- Become dense and visible in CT scans when heart muscle cells die.
Answers: C,A
