Please click here to go to the most recent version of this course
Modes and mechanisms of transmission of pathogenic organisms in the healthcare setting and strategies for prevention and control.
It is becoming increasingly clear that transmission of infections in healthcare settings is largely preventable through the use of evidence-based infection control guidelines. The concept of the chain of infection has provided the basis for understanding the transmission of pathogens as well as identifying practices and procedures to prevent healthcare-associated infections.
A healthcare-associated infection (HAI) is defined as a localized or systemic condition that (1) results from an adverse reaction to the presence of an infectious agent or its toxin, (2) occurs during a hospital admission, (3) has no evidence of the infection being present or incubating at admission, and (4) meets body site-specific criteria.
Antibiotic-resistant organisms have changed the infection control landscape. Methicillin-resistant S. aureus (MRSA), C. difficile, and vancomycin-resistant enterococcus (VRE), among others, have become serious problems in healthcare facilities over the past decades.
Staphylococcus aureus
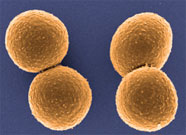
This colored electron micrograph shows isolated S. aureus bacteria that are resistant to many forms of antibiotics. Source: NIAID.
MRSA
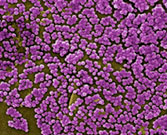
Scanning electron micrograph depicting numerous clumps of MRSA bacteria. Source: CDC. Public domain.
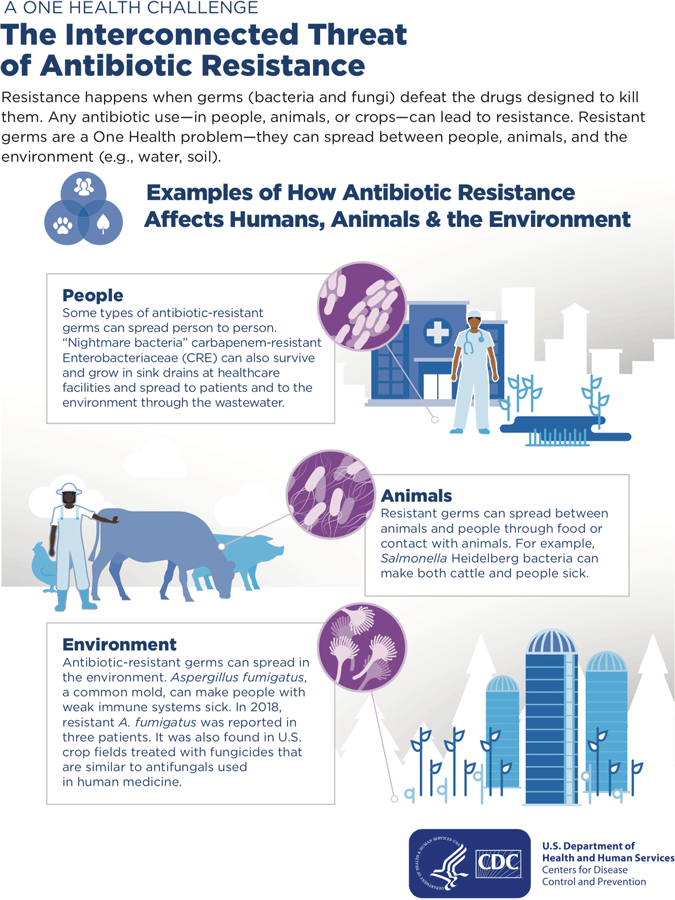
Microbe Resistance
Antimicrobial resistance (AMR) threatens the effective prevention and treatment of an ever-increasing range of infections caused by bacteria, parasites, viruses and fungi.
Antimicrobial resistance happens when microorganisms (such as bacteria, fungi, viruses, and parasites) change when they are exposed to antimicrobial drugs (such as antibiotics, antifungals, antivirals, antimalarials, and anthelmintics). Microorganisms that develop antimicrobial resistance are sometimes referred to as “superbugs.”
As a result, the medicines become ineffective and infections persist in the body, increasing the risk of spread to others.
Antibiotic resistance occurs when bacteria change in response to the use of these medicines. Bacteria, not humans or animals, become antibiotic-resistant. These bacteria may infect humans and animals, and the infections they cause are harder to treat than those caused by non-resistant bacteria.
Examples of bacteria that are resistant to antibiotics include methicillin-resistant Staphylococcus aureus (MRSA), penicillin-resistant enterococcus, and multidrug-resistant Mycobacterium tuberculosis (MDR-TB), which is resistant to two tuberculosis drugs, isoniazid and rifampicin.
Source: WHO, 2020.
Clostridium difficile has also become more virulent, and hospital-associated outbreaks are causing increased deaths. In the general population, C. difficile is present in about 5% of the population. The need to control outbreaks of C. difficile has focused new attention in the area of environmental cleaning. Because C. difficile causes watery diarrhea it can spread easily and rapidly in the healthcare setting, passing from person to person via clothing, equipment, and dirty hands.
Vancomycin-resistant enterococcus (VRE) is another antibiotic-resistant organism that has been associated with increased mortality and length of hospital stay. Many studies have shown that VRE can be readily found on cabinets, bedrails, equipment, and bedside tables and it is easily transmitted on the hands, gloves, and clothing of healthcare workers. Vancomycin-resistant enterococcus is also easily transmitted on equipment such as blood pressure cuffs, stethoscopes, pulse oximeters, IV poles, telephones, and infusion pumps. Aggressive environmental cleaning, screening of incoming patients for VRE and MRSA, isolation, and stringent barrier precautions have led to remarkable success in controlling and eliminating these organisms in some areas.
Source: CDC, 2020a.
The Influenza Virus
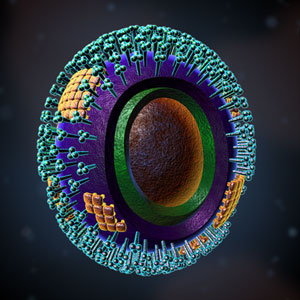
Source: Zygote Media. Used with permission.
Even before the recent H1N1 outbreak, influenza has long been an area of focus for infection prevention. Although flu pandemics have occurred periodically for centuries, causing hundreds of thousands of deaths, we now have the ability to identify an influenza epidemic as it is emerging. We also have the public health capability to take action (vaccines, surveillance, education) to minimize the impact of a flu outbreak. Addressing and controlling these emerging threats has become a priority for healthcare organizations.
The Chain of Infection
The Chain of Infection
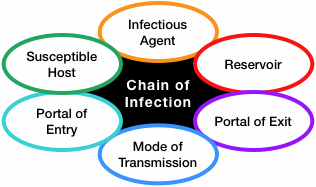
Source: Author.
We have all seen infections spread through a family, classroom, or office; this situation can be described using a concept called the chain of infection (see figure). It is a process that begins when (1) an infectious agent or pathogen (2) leaves its reservoir, source, or host through (3) a portal of exit, (4) is conveyed by some mode of transmission, (5) enters the host through an appropriate portal of entry, and (6) infects a susceptible host. The now-infected susceptible host becomes a new reservoir and the whole process starts over.
The concept of a chain of infection is essential to our understanding of why we do what we do to prevent infection. If any link of the chain of infection can be broken, the spread of infection can be prevented.Infectious Agents or Pathogens
Infectious agents or pathogens are the microorganisms or “germs”—bacteria, viruses, fungi, and protozoa—that can cause disease or illness in its host. Some microorganisms are pathogens, a word derived from the Greek, meaning “that which produces suffering.” Although microorganisms are common in the environment, most are not harmful to people.
Pathogens vary in infectivity and virulence, and to cause disease an infectious dose (a sufficient number of organisms) is required. Creating an environment with no pathogens is not a realistic goal outside of highly specialized laboratories.
Bacteria
Bacteria are single-celled organisms, the vast majority of which are harmless or even beneficial. Our bodies always contain bacteria, called normal flora, that protect us from infection by providing competition to pathogens. Normal flora usually do not cause disease unless balance is disturbed or the bacteria get into a part of the body that cannot tolerate them. Antibiotics are effective against many bacterial infections although, as already noted, the overuse or misuse of antibiotics has produced strains of bacteria that are resistant to them.
Pathogenic bacteria contribute to a number of globally prevalent diseases, including pneumonia, tuberculosis, and bacterial meningitis. Pathogenic bacteria include group A and group B streptococcus; Haemophilus influenzae; Staphylococcus aureus, including MRSA; Clostridium difficile; Neisseria meningitidis; and Streptococcus pneumoniae.
Coccus
A coccus is a bacterium with a spherical shape. Chains of cocci indicate streptococcus, while clusters indicate staphylococcus. Source: Zygote Media. Used with permission.
Bacillus

Bacillus can refer to any rod-shaped bacterium, or can be more specific to Bacillus, which is a gram-positive and rod-shaped genus. Source: Zygote Media. Used with permission.
Viruses
Novel Coronavirus 19, or COVID-19
Source: CDC, 2020. Public domain.
Viruses are true parasites in that they can only reproduce inside the host cell. More than 5,000 types of viruses have been described since the first was discovered in 1899, the newest being COVID-19, which has sparked a pandemic worldwide in 2020. Viruses are about a hundred times smaller than bacteria and, like bacteria, not all viruses cause disease.
Viruses spread in many different ways—by direct or indirect contact (soiled hands or articles), by droplets from coughing and sneezing, by contact with blood, sexual contact, fecal contamination, contaminated food and water, or via certain insects. Examples of diseases caused by viruses include influenza, chickenpox, West Nile fever, HIV and COVID-19.
Antibiotics are not effective against viruses. Vaccines, however, have been successful in eliminating or controlling some viral disease—including smallpox, polio, measles, mumps, and rubella—that have killed millions of people throughout the world. Anti-viral medications for some illnesses have varying degrees of effectiveness. Countries worldwide were caught unprepared for the novel virus that was identified as coronavirus 19.
West Nile Virus
This image of the West Nile Virus shows the characteristic rough and furrowed surface with no protein arms projecting from it, as so many viruses have. Source: Zygote Media. Used with permission.
Human Immunovirus (HIV)
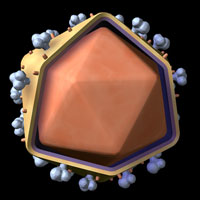
HIV is a retrovirus, whose genetic content is stored in RNA, which is copied into the DNA of the host upon infection. Source: Zygote Media. Used with permission.
Fungi
Ringworm Infection
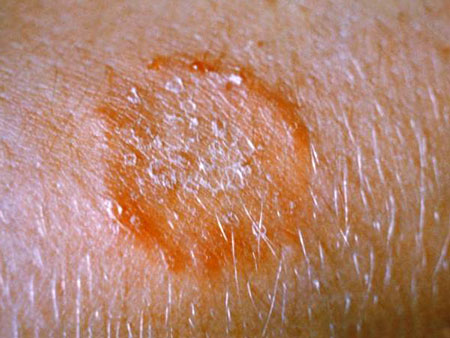
An example of a fungal infection called ringworm (no worm is involved). Source: CDC. Public domain.
Fungi are very common, but only a few cause diseases in humans. Some fungal infections are life-threatening in certain susceptible patients. Fungal infections can be superficial (limited to the surface of the skin and hair), cutaneous (extending into the epidermis, nails, and hair), or subcutaneus (extending into the dermis, subcutaneous tissues, muscle, and fascia). Fungal infections can also be systemic, often originating in the lungs and spreading to multiple organs. There are several classes of antifungal medications, although fungal and human cells are similar on the molecular level, so antifungal drugs can have mild to serious side effects. Athlete’s foot, yeast infections, and candidemia (yeast growing in the blood) are examples of diseases caused by fungi.
One fungus that survives well in air, dust, and moisture in healthcare facilities is Aspergillus spp., a ubiquitous, aerobic fungus that is present in soil, water, and decaying vegetation. Site renovation and construction can disturb Aspergillus-contaminated dust and produce bursts of airborne fungal spores, which have been associated with clusters of HAIs in immunocompromised patients. Absorbent building materials such as wallboard are an ideal growth medium for this organism if they become and remain wet. Patient-care items, devices, and equipment can become contaminated with Aspergillus spp. spores and serve as sources of infection (CDC, 2003, 2019).
Other opportunistic fungi that are occasionally linked with HAIs are members of the order Mucorales and molds such as fusarium and penicillium. Many of these fungi can proliferate in moist environments‑as noted before in water-damaged wood and building materials. Some fungi, such as fusarium and pseudoallescheria, can be airborne. As with aspergillosis, a major risk factor for disease caused by any of these pathogens is the host’s severe immunosuppression from either underlying disease or immunosuppressive therapy (CDC, 2003, 2019).
Protozoa
Protozoa are single- or multi-celled microorganisms that are larger than bacteria. They have traditionally been classified by their means of propulsion: flagella, amoeboid, sporozoan, or ciliate. They may be transmitted in soil, via water, by direct contact, or by an insect such as a mosquito.
Examples of diseases caused by protozoa include malaria and giardia. Malaria is a protozoan which lives in the blood of the host and is transmitted when an insect bites, ingests infected blood, and then transmits it by biting a new host.
Protozoa are less common than the other types of organisms in the United States and can be treated with specific medications.
Two Views of Giardia
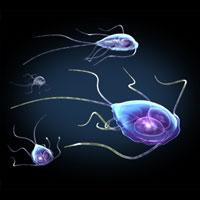
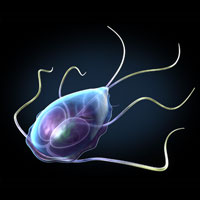
These images depict Giardia trophozoites in a variety of positions. Giardia stick closely to the lining of the small intestine in the hosts they infect and cause mild to severe diarrhea. Source: Zygote Media. Used with permission.
Parasites
Parasites are usually larger organisms that exploit a host by living on the skin, inside the gut, or in tissues. The life of a parasite is precarious because the host usually does everything it can to destroy the parasite. Parasites are dependent upon the host for survival and employ a number of strategies to move from host to host. They can be transmitted by direct contact, as with lice or scabies, or wait in the external environment until there is contact with the host (ticks, leeches).
Helminthes are a class of parasites that live inside the body and include roundworms, tapeworms, and flukes. They infect humans principally through ingestion of fertilized eggs or when the larvae penetrate the skin or mucous membranes.
Ringworm
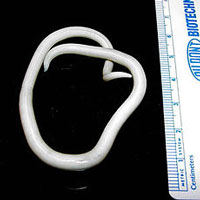
The parasitic roundworm Ascaris lumbricoides. As many as one-quarter of the world’s population is infected with Ascaris. Source: Wikipedia. Used by permission.
Louse

Pediculus humanus var capitis, also known as head louse. Source: Wikipedia. Used by permission.
Reservoirs
Reservoirs are the places where the germs live and grow. A general rule: If an area stays wet, it is probably a reservoir. The most common reservoirs in healthcare facilities are people, who may be sick or healthy.
Infectious agents transmitted during healthcare derive primarily from human sources. Human reservoirs include patients, healthcare personnel, household members, and visitors. These source individuals may have active infections, may be in the asymptomatic or incubation period of an infectious disease, or may be transiently or chronically colonized with pathogenic microorganisms, particularly in the respiratory and gastrointestinal tracts (CDC, 2007, 2019).
Surprisingly, reservoirs can be complex and difficult to identify. The CDC defines a reservoir as “one or more epidemiologically connected populations or environments in which the pathogen can be permanently maintained and from which infection is transmitted to the defined target population.”
Haydon and colleagues (2002) stated that
. . .any infectious agents, especially those that cause emerging diseases, infect more than one host species. Managing reservoirs of multi-host pathogens often plays a crucial role in effective disease control. However, reservoirs remain variously and loosely defined. We propose that reservoirs can only be understood with reference to defined target populations. Therefore, we define a reservoir as one or more epidemiologically connected populations or environments in which the pathogen can be permanently maintained and from which infection is transmitted to the defined target population.
Existence of a reservoir is confirmed when infection within the target population cannot be sustained after all transmission between target and nontarget populations has been eliminated. When disease can be controlled solely by interventions within target populations, little knowledge of potentially complex reservoir infection dynamics is necessary for effective control. (Haydon et al., 2002).
In humans, the reservoir and the susceptible host can be the same person and can cause disease if the person’s normal flora gets into the wrong part of the body. For example, oral flora getting into the lungs can cause aspiration pneumonia, skin flora contaminating an IV site can cause a site or bloodstream infection, and fecal flora contaminating the urinary tract can cause a urinary tract infection (UTI). This is why care must be taken to avoid carrying germs across different body sites of the same patient. The most effective prevention technique is to change gloves and do hand hygiene when going from a contaminated area to a cleaner area.
In healthcare facilities, activities aimed at eliminating reservoirs include:
- Treating people who are ill
- Handling and disposing of body fluids carefully
- Using sterile water in respiratory equipment
- Drying equipment before storing it
- Handling food safely and cooking meat thoroughly
- Monitoring soil and contaminated water in sensitive areas of the hospital and washing hands carefully after contact with either
- Vaccinating people
- Encouraging ill workers to stay home
Infection control practices should be followed in all settings where healthcare is delivered, including home care, although the relative risk of acquiring an infection may differ. In acute care, a patient’s risk for an HAI is related not only to the severity of illness and exposure to invasive interventions and devices but also to environmental risks, including exposure to other patients and inanimate reservoirs or pathogens.
In home care, the rationale and strategy for use of precautions differ from those applied in hospitals. In most cases, the use of gowns, gloves, and masks in the care of homebound patients is recommended to protect the healthcare provider, not the patient.
Home care patients known to have a multidrug-resistant organism should be cared for using appropriate barriers. Although these organisms may not be a risk to providers, they may be transmitted to other homecare patients through inanimate objects or hands. Reusable equipment such as stethoscopes and blood pressure cuffs should remain in the home.
For guidance regarding home-based patients with COVID-19, click here.
Portals of Exit: How Pathogens Leave the Body
A pathogen leaves its reservoir or host through a portal of exit. The portal of exit usually corresponds to the site where the pathogen is located. For example, influenza viruses and M. tuberculosis exit from the respiratory tract, cholera exits its host in feces, and Sarcoptes scabiei in scabies skin lesions. Some bloodborne pathogens can exit by crossing the placenta from mother to fetus (rubella, syphilis, toxoplasmosis), while others exit through cuts in the skin or needles (hepatitis B) or blood-sucking insects (malaria).
The portal of exit is the link of the chain over which we have the least control. Any break in the skin—such as natural anatomical openings or draining lesions—may be a portal of exit from a host. Any body fluid may carry infectious agents out of the body. Some bacteria (such as MRSA) live on the patient’s skin, so even dry skin contact may serve as the portal of exit.
Activities aimed at eliminating portals of exit in healthcare facilities include:
- Covering coughs and sneezes with a tissue
- Handling body fluids with gloves—followed by hand hygiene
- Keeping draining wounds covered with a dressing
- Staying home from work when you have wet lesions or weeping dermatitis
Means of Transmission
Very few germs can fly—almost all have to be carried from one place to another. The means of transmission is the weakest link in the chain of infection, and it is the only link we can hope to eliminate entirely. Most infection control efforts are aimed at preventing the transport of germs from the reservoir to the susceptible host.
All types of precautions (standard, contact, droplet, and airborne) are designed to interrupt the means of transmission. These are reviewed in detail under “Prevention Strategies.” Direct and indirect contact are the most common means of transmission in the healthcare setting—from the hands of the caregivers and items that move patient to patient. Because it addresses the weakest link in the chain of transmission,
Hand hygiene is still the single most important procedure for preventing the spread of infection.
Items moving between patients should be cleaned after each use to avoid indirect contact transmission of pathogens.
Common Means of Transmission | |
|---|---|
Type of contact | Example |
Direct | Person-to-person transmission of pathogens through touching, biting, kissing, or sexual intercourse |
Indirect | Involves an intermediate person or item between the portal of exit and the portal of entry to the next person. Microorganisms may be carried by unwashed hands or soiled objects, called fomites. Any soiled object, such as blood-pressure cuffs, pens, bed rails, used tissues, soiled laundry, or doorknobs, may be a fomite. |
An agent or pathogen can be indirectly transmitted from a reservoir to a susceptible host on inanimate objects. Cleaning and disinfection are important practices to ensure that medical equipment surfaces do not serve as reservoirs for infectious pathogens. Hands of healthcare personnel may transmit pathogens after touching an infected or colonized body site on a patient or a contaminated inanimate object if hand hygiene is not performed before touching another patient (CDC, 2003, 2019).
Patient-care devices, such as electronic thermometers, glucose monitoring devices, stethoscopes, blood-pressure cuffs, and other devices may transmit pathogens if they are contaminated with blood or bodily fluids or are shared between patients without cleaning and disinfecting. Shared toys may become a vehicle for transmitting respiratory viruses (e.g., respiratory syncytial virus) or pathogenic bacteria (e.g., Pseudomonas aeruginosa) among pediatric patients (CDC, 2003, 2019).
Toys used by young children should be washable. A system should ensure that they are washed and dried routinely. Older children should wash hands before and after using shared toys or equipment.
Instruments (e.g., endoscopes, surgical instruments) that are inadequately cleaned between patients before disinfection or sterilization or that have manufacturing defects that interfere with the effectiveness of reprocessing may transmit bacterial and viral pathogens. Clothing, uniforms, laboratory coats, or isolation gowns used as personal protective equipment (PPE) may become contaminated with potential pathogens after care of a patient colonized or infected with an infectious agent (CDC, 2003, 2019).
The potential also exists for soiled garments to transfer infectious agents to successive patients. A 2007 study in a Maryland teaching hospital revealed that 27% of the white coats worn by 109 doctors and other medical professionals were colonized with S. aureus and 6% were colonized with MRSA. In a followup questionnaire, 65% of the healthcare workers reported they had last washed their white coat more than a week ago and nearly 16% had last washed their coat more than 30 days ago (Treakle et al., 2009).
Transmission of germs can also occur through the air via droplet or airborne routes. Droplet transmission is common, easily spreading infections such as colds, influenza, whooping cough (pertussis), and some forms of meningitis. Droplets are produced when the infected person coughs, sneezes, or speaks. Droplets can travel about 3 to 6 feet before drying out or falling to the ground. Droplet Precautions are designed to interrupt this means of transmission, and respiratory hygiene practices recommend that they be used when caring for any person with active respiratory symptoms.
A Demonstration of Droplet Transmission

This photograph captures a sneeze in progress, revealing the plume of salivary droplets as they are expelled in a large cone-shaped array from this man’s open mouth, thereby dramatically illustrating the reason for covering your mouth when coughing or sneezing in order to protect others from germ exposure. COVID-19 is spread primarily in this way. Source: CDC.
Airborne transmission occurs with only a few infections —those caused by organisms that can survive the drying of respiratory droplets. When the droplets evaporate, they leave behind droplet nuclei, which are so tiny they remain suspended in the air. Diseases transmitted by the airborne route include tuberculosis, chickenpox, measles, severe acute respiratory syndrome (SARS), smallpox, and COVID-19. Airborne Precautions are designed to interrupt this means of transmission.
Means of transmission that are not common in hospitals include:
- Common-source vehicles such as contaminated food, water, milk, or IV fluid. In hospitals, these products are obtained only from safe and approved sources to prevent contamination.
- Vector-borne transmission by an animal carrier such as a rat or mosquito that carries the pathogen from reservoir to host. Hospitals maintain their environment so that vector-borne transmission is not likely to occur.
Activities aimed at eliminating the means of transmission in healthcare facilities include:
- Hand hygiene
- Wearing gloves to minimize contamination of hands and discarding them after each patient
- Maintaining Standard, Contact, Droplet and Airborne Precautions as indicated
- Cleaning, disinfection, or sterilization of equipment used by more than one patient
- Cleaning of the environment, especially high-touch surfaces
- Maintaining directional air flow
Portals of Entry: How Pathogens Are Introduced
The portal of entry refers to the location through which a pathogen enters a susceptible host. The portal of entry must provide access to tissues in which the pathogen can multiply or a toxin can act. Often, the infectious agent uses the same portal to enter the new host that it used to exit the source host. For example, influenza virus exits the respiratory tract of the source host and enters the respiratory tract of the new host.
Other pathogens follow a so-called fecal–oral route because they exit the source host in feces, are carried on inadequately washed hands to a vehicle such as food, water, or utensils, and enter a new host through the mouth. Other portals of entry include skin, mucous membranes, and blood.
Pathogens cannot cause illness until they gain entry into the body, and, in general, they cannot enter through intact skin. They may gain entry through an anatomic opening, a skin break caused by illness or accident, or an opening created during a medical procedure, such as a surgical wound or an IV site. Preventing or eliminating portals of entry, where possible, and protecting portals that cannot be eliminated is a must for both patients and healthcare personnel.
Examples of portals of entry include:
- Mouth, nose, eyes, and other anatomic openings
- Rash or dermatitis
- Insect bites
- Injuries, from microscopic to major
- Surgical wounds
- Intravenous sites
- Any location, whether anatomic or created, with a tube in place
- Needle-puncture injuries
Activities aimed at protecting or eliminating portals of entry in healthcare facilities include:
- Use of aseptic surgical technique
- Application of dressings on surgical wounds
- Use of IV site dressings and proper care
- Elimination of tubes as soon as possible
- Use of masks, goggles, and face shields
- Protecting your skin to prevent holes (such as dermatitis)
- Keeping unwashed hands and objects away from the mouth
- Use of actions and devices to prevent needlesticks
Susceptible Host
The final link in the chain of infection is the susceptible host. Most of the factors that influence infection and the occurrence and severity of disease are related to the host, although agent and environmental factors also play a role (table below). However, characteristics of the host-agent interaction—such as pathogenicity, virulence, and antigenicity—are also important. The infectious dose, mechanism of disease production, and route of exposure are also factors.
Factors that Influence the Outcome of an Exposure | |
|---|---|
Host factors |
|
Agent factors |
|
Environmental factors |
|
Some people exposed to pathogenic microorganisms never develop symptomatic disease while others become severely ill and even die. Those who are extremely old or young, are already ill, have holes in their skin, have invasive devices in place, or are immunocompromised are more susceptible. Still others progress from colonization to symptomatic disease either immediately following exposure or after a period of asymptomatic colonization.
Susceptibility can be reduced in several ways. For some diseases there are effective vaccines and some diseases produce lasting immunity after illness. We have better resistance to disease when we are well rested, well fed, and relatively stress-free. People with healthy immune systems are often able to resist infection even when bacteria do invade.
The healthy body has numerous protective structures and systems that support resistance to infection. These include intact skin, blood circulation bringing white blood cells and nutrients to the tissues, antibodies to previously encountered infectious agents, the inflammatory response, stomach acid, and a robust community of normal flora, which provides competition to invading pathogens. A person with these defense mechanisms intact is said to be immunocompetent.
Immunocompromise varies in severity and can be temporary or long term. A person who is sick in bed for a few days may be mildly compromised, while a person with a chronic illness such as diabetes is probably moderately and chronically compromised. Someone receiving chemotherapy or a transplant patient may be severely immunocompromised.
Extra care should be taken to protect a person who is immunocompromised. Nutritional status should be closely monitored to support immune competence. The care should be tailored to the specific needs and situation of the patient. Both the very young and very old need extra protection from infection. Any indwelling device (e.g., IV catheters, urinary catheters) increases susceptibility. To reduce the risk of infections associated with these devices, the device should be discontinued as soon as the patient no longer needs it.
Infections are sometimes more related to host factors than to the infectious agent. For example, a person who is well rested may resist the virus that makes the over-tired person sick. Some organisms are widely found but only cause disease in a susceptible host—such as the person recently treated with antibiotics who then develops a yeast infection. Examples of susceptible hosts include people who:
- Are already ill
- Have invasive devices or tubes in place
- Are malnourished
- Are very old or very young
- Are tired or under high stress
- Have skin breaks such as surgical wounds or IV sites
- Are undergoing steroid therapy or treatment for cancer
- Have HIV infection
- Are well and healthy! (No one is immune to all disease.)
Activities aimed at protecting or eliminating susceptible hosts in healthcare facilities include:
- Preventing exposure of both patients and staff to communicable disease
- Removal of invasive devices as soon as they are no longer needed
- Maintaining good nutrition
- Maintaining good skin condition
- Covering skin breaks
- Vaccinating people against illnesses to which they may be exposed
- Encouraging rest and balance in our lives
Prevention Strategies
[Unless otherwise cited, the material for this section was take from CDC, 2003, 2019.]
Both science and regulation address prevention of healthcare-associated infections. The CDC provides the chief authority for science. Regulations may be federal, state, or local.
Since 1991, when OSHA first issued its Bloodborne Pathogens Standard to protect healthcare personnel from blood exposure, the focus of regulatory and legislative activity has been on implementing a hierarchy of prevention and control measures. A central tenet is to consider all patients to be potentially infected with a bloodborne pathogen.
The federal OSHA Bloodborne Pathogens Standard requires that each employer having employees with occupational exposure to blood or other potentially infectious material (OPIM) shall establish a written exposure control plan designed to eliminate or minimize employee exposure. Among other things, this plan must address:
- Standard/Universal Precautions, including hand hygiene (Element II)
- Engineering and work practice controls (See Element III.)
- Personal protective equipment (PPE) (See Element IV.)
- Housekeeping, Laundry, regulated waste (See Element V.)
- Contaminated sharps and equipment (See Element III.)
- Hepatitis B vaccination and exposure follow-up (See element VI.)
- Employee communication and education (See Element VI.)
- Recordkeeping
The Exposure Control Plan must be available to employees. Many of the educational requirements are addressed in this course, but it does not take the place of an employer-specific Exposure Control Plan.
The complete federal Bloodborne Pathogens Standard is available here.
Standard Precautions
Initially, Universal Precautions were developed by OSHA to protect healthcare workers from bloodborne pathogens, such as HIV, Hepatitis B (HBV), and hepatitis C (HCV). Universal Precautions were developed for use with all patients because those with bloodborne infections may be asymptomatic or unaware of their infectious status. Universal Precautions continue to be required by the OSHA Bloodborne Pathogens Standard, although they have been subsumed into the more comprehensive Standard Precautions (see below).
Universal Precautions requires avoidance of contact with blood or other potentially infectious materials (OPIM). OSHA defines “other potentially infectious materials” as (1) The following human body fluids: semen, vaginal secretions, cerebrospinal fluid, synovial fluid, pleural fluid, pericardial fluid, peritoneal fluid, amniotic fluid, saliva in dental procedures, any body fluid that is visibly contaminated with blood, and all body fluids in situations where it is difficult or impossible to differentiate between body fluids; (2) Any unfixed tissue or organ (other than intact skin) from a human (living or dead); and (3) HIV-containing cell or tissue cultures, organ cultures, and HIV- or HBV-containing culture medium or other solutions; and blood, organs, or other tissues from experimental animals infected with HIV or HBV.
Because the focus of Universal Precautions was narrow (to protect healthcare workers from bloodborne pathogens), the CDC was led to develop Standard Precautions, which include all of Universal Precautions and more.
Note
When we use Standard Precautions, we are in full compliance with Universal Precautions.
Standard Precautions protect patients and healthcare workers from many bacterial and viral infections, including bloodborne pathogens.
Recommended Use of Standard Precautions for All Patients, All Settings | |
|---|---|
Component | Recommendations |
Hand hygiene | After touching blood, body fluids, secretions, excretions, contaminated items; immediately after removing gloves; between patient contacts. |
Personal protective equipment (PPE) | |
Gloves | For touching blood, body fluids, secretions, excretions, contaminated items; for touching mucous membranes and nonintact skin |
Gown | During procedures and patient-care activities when contact of clothing/exposed skin with blood/body fluids, secretions, and excretions is anticipated |
Mask, eye protection (goggles), face shield* | During procedures and patient-care activities likely to generate splashes or sprays of blood, body fluids, secretions, especially suctioning, endotracheal intubation |
| |
Soiled patient-care equipment | Handle in a manner that prevents transfer of microorganisms to others and to the environment; wear gloves if visibly contaminated; perform hand hygiene. |
Environmental control | Develop procedures for routine care, cleaning, and disinfection of environmental surfaces, especially frequently touched surfaces in patient-care areas. |
Textiles and laundry | Handle in a manner that prevents transfer of microorganisms to others and to the environment. |
Needles and other sharps | Do not recap, bend, break, or hand-manipulate used needles; if recapping is required, use a one-handed scoop technique only; use safety features when available; place used sharps in puncture-resistant container. |
Patient resuscitation | Use mouthpiece, resuscitation bag, other ventilation devices to prevent contact with mouth and oral secretions. |
Patient placement | Prioritize for single-patient room if patient is at increased risk of transmission, is likely to contaminate the environment, does not maintain appropriate hygiene, or is at increased risk of acquiring infection or developing adverse outcome following infection. |
Respiratory hygiene/cough etiquette (source containment of infectious respiratory secretions in symptomatic patients, beginning at initial point of encounter e.g., triage and reception areas in emergency departments and physician offices) | Instruct symptomatic persons to cover mouth/nose when sneezing/coughing; use tissues and dispose in no-touch receptacle; observe hand hygiene after soiling of hands with respiratory secretions; wear surgical mask if tolerated or maintain spatial separation, >3 feet if possible. |
Standard Precautions tell us to avoid contact with:
- Blood and all body fluids from all patients. Any body fluid may carry microorganisms.
- Mucous membranes
- Non-intact skin (abrasions, dermatitis, rash)
Standard Precautions, as described by the CDC, are related to the Elements that organize our understanding of infection control:
- Assume that every person is potentially infected or colonized.
- Use personal protective equipment (PPE)—see Element IV. Note that mask, eye protection, and gown may be required for care of a patient on Standard Precautions. Supplies must be readily available.
- Implement Respiratory Hygiene in all areas where people with respiratory symptoms may be seen.
- Ensure appropriate patient placement: follow isolation precautions.
- Clean patient-care equipment and the environment as described in Element V.
- Handle textiles and laundry carefully.
- Perform hand hygiene correctly and routinely.
- Use safe injection practices to protect patients and workers (follow sharps safety)—Elements II and III. (CDC, 2016).
Respiratory hygiene was incorporated into Standard Precautions by the CDC (2007, updated in 2019), to:
- Educate healthcare personnel on the importance of source control measures to prevent droplet and fomite transmission of respiratory infection. Cover the portal of exit!
- Put these measures in place beginning at the point of initial encounter in the healthcare setting.
- Apply them to both patients and accompanying individuals.
- Post signs at entrances and other key locations asking people with respiratory symptoms to cover their mouths and noses when coughing or sneezing, to dispose of tissues and do hand hygiene. Signs in several languages are available from the CDC.
- Provide tissues, waste receptacles, and hand hygiene materials.
- Offer a simple mask to coughing patients.
- Encourage coughing patients to stay at least 3 feet away from others.
Correct Use of Standard Precautions | |
|---|---|
Always |
|
Never |
|
Transmission-Based Precautions
The CDC also recommends, for patients with certain infections, use of transmission-based precautions, in addition to Standard Precautions. Standard Precautions are used with all patients and do not require a sign on the door. Patients being cared for using Contact, Droplet, or Airborne Precautions will have a sign on the door in most facilities. Note that the sign on the door may not specify the patient’s diagnosis for reasons of privacy.
Precautions may vary between facilities. Refer to your facility’s policies for details. Details for all types of precautions may be found here.
Facilities should have policies for transport of the patient outside the room, addressing each type of transmission-based precautions.
Contact Precautions
The following are CDC recommendations for acute-care facilities. Other types of facilities should develop policies based on the Guideline.
- Hand hygiene is critical in the care of these patients.
- Visitors must perform hand hygiene on leaving the room.
- Use a single-patient room as possible. Consult with Infection Control staff if not possible.
- Wear gloves to enter the room. Change gloves as specified by Standard Precautions.
- Wear a gown to enter the room.
- Use single-patient equipment, left in the room, as possible.
- Disinfect any equipment that must leave the room.
- Clean and disinfect these rooms at least daily.
Supplies needed include:
- Gloves
- Gowns
- Disinfectant for removed equipment
- Trash container for discarded PEP
- Single-patient use equipment
Contact Precautions are often used to care for patients with Methicillin-resistant Staph aureus, C. difficile, wounds with uncontained drainage, and a number of other infections.
Droplet Precautions
The following are CDC recommendations for acute-care facilities. Other types of facilities should develop policies based on the Guideline.
- Use a single-patient room as possible. Consult with Infection Control staff if not possible.
- Maintain at least 3 feet separation between the patient and others.
- If two patients must share a room, draw the privacy curtain between them.
- Hand hygiene must be done as specified for Standard Precautions.
- Staff should wear a simple mask to enter the room.
- Supplies needed (beyond Standard Precautions) are limited to simple masks.
Droplet Precautions are used to provide care to patients with influenza, pertussis, some types of meningitis, undiagnosed respiratory infections, and several other diseases.
Airborne Precautions
Airborne Precautions are the only type that require:
- Airborne Infection Isolation room—AIIR—a negative pressure isolation room
- N-95 respirator or PAPR. See the discussion of tuberculosis, following, for these types of respiratory protection.
- Supplies needed (beyond Standard Precautions) are limited to the appropriate respiratory protection.
AIIRs have very specific requirements and are often available only in acute care facilities. If a disease requiring Airborne Precautions is suspected and an AIIR is not available, place a simple mask on the patient and place him/her in a separate room with its door closed while transfer to a facility with an available AIIR is arranged. Non-acute care settings should have well-known policies for identifying and managing such patients.
If patients must come out of the AIIR, put a simple mask on them, as a tight-fitting respirator may not be tolerated and is not indicated.
Airborne Precautions are used for patients known or suspected of having:
- Tuberculosis, active pulmonary or rule-out
- Chickenpox
- Measles
- Disseminated herpes zoster (shingles of more than one dermatome)
- SARS
- Smallpox
- COVID-19
In 2019 reported occurrences of tuberculosis in the United States were 8,920 (CDC, 2019b), and worldwide a total of 1.5 million was reported for 2018(WHO, 2019). Tuberculosis is a bacterial infection caused by Mycobacterium tuberculosis and is spread in airborne droplets when people with the disease cough or sneeze. Most people with healthy immune systems infected with M. tuberculosis never become ill. However, the bacteria remain dormant within the body and can cause tuberculosis years later if host immunity declines.
The person who is most likely to transmit tuberculosis is the person who has not been diagnosed —the unknown carrier. Identification without delay of the person with active tuberculosis is critical so that isolation and treatment can prevent transmission to others.
Active TB does have symptoms, which depend on where in the body the TB bacteria are growing. Tubercular disease in the lungs may cause symptoms such as a bad cough that lasts 3 weeks or longer, pain in the chest, or coughing up blood or sputum (phlegm from deep inside the lungs). Other symptoms of active TB disease are weakness or fatigue, weight loss, no appetite, chills, fever, or sweating at night.
Diagnostic tests for the disease include chest x-rays, the tuberculin skin test, and sputum cultures. Active disease takes 6 to 9 months to treat because it takes that long for all the TB bacteria to die. The most common medications used to treat TB disease are isoniazid, rifampin, ethambutol, and pyrazinamide.
Because tuberculosis is the primary disease transmitted by a true airborne route, and because it is the undiagnosed person who is most likely to transmit disease, the CDC recommends a three-level hierarchy of controls: administrative, environmental, and respiratory protection controls.
Administrative controls specify who is in charge of the facility’s TB control program, including critical infrastructure such as laboratories as well as other services needed to maintain an effective program. A key component is having a plan to ensure prompt detection, airborne precautions, and treatment of persons who have suspected or confirmed TB disease. Diagnose, isolate, and treat to prevent exposing others. Environmental controls are responsible for containing the source of exposure, primarily by the use of Airborne Infection Isolation (AII) rooms that provide negative-pressure ventilation.
Powered Air-purifying Respirator (PAPR) Hood
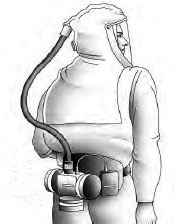
Source: CDC. Public domain.
Respiratory controls address the protection of people who must be protected from contaminated air when they enter the AII room. Most facilities provide N-95 respirators, which must be fit-tested. Some facilities exclusively use powered air-purifying respirators (PAPRs, see below) for all staff; they do not require fit testing. Check your facility’s policies for what respiratory protection is made available for visitors.
Tuberculosis infectiousness usually declines within weeks of beginning treatment. The patient must show clear clinical improvement before isolation is discontinued because the patient with resistant organisms remains infectious if initial treatment is not effective. Airborne Precautions for tuberculosis may be discontinued when both of the following criteria have been met: (1) clinical improvement, and (2) three consecutive sputum smears negative for acid-fast bacilli (TB germs).
Each of the three sputum specimens should be collected in eight 24-hour intervals and at least one specimen should be an early morning specimen.
For current guidelines, consult CDC Guidelines for Preventing the Transmission of Mycobacterium tuberculosis in Health-Care Settings (CDC, 2003, 2019).
Multidrug-resistant TB (MDR-TB) and extensively drug-resistant tuberculosis (XDR-TB) have become more common and are highly infectious. Treatment of drug-resistant TB is much more difficult than normal tuberculosis, requiring even more antibiotics, and for 20–30 months and beyond (WebMD.com, 2019).
Hand Hygiene
The term hand hygiene includes both the use of an alcohol-based hand rub and washing with soap and water.
The chain of infection makes it clear why hand hygiene is critical. For generations, handwashing with soap and water has been the standard measure of personal hygiene. The concept of cleansing hands with an antiseptic agent probably emerged in the early nineteenth century. As early as 1822, a French pharmacist demonstrated that solutions containing chlorides of lime or soda could eradicate the foul odors associated with human corpses and that such solutions could be used as disinfectants and antiseptics. In a paper published in 1825 this pharmacist stated that physicians and other persons attending patients with contagious diseases would benefit from moistening their hands with a liquid chloride solution.
2002 CDC Guidelines for Hand Hygiene brought a major change in hand hygiene practices. While washing with soap and water is still required in some situations, now the use of an alcohol-based hand rub is preferred for routine use.
Despite the simplicity and effectiveness of hand hygiene in preventing the spread of infectious disease, adherence to hand hygiene practice remains unacceptably low throughout the world. Although measuring hand hygiene adherence is not a simple task, an oft-cited study by Pittet (2001) noted that adherence varies among professional categories of healthcare workers and between hospital departments but is usually estimated as less than 50%.
CDC (2002) has described observed and self-reported factors that influence adherence to hand hygiene practices.
Observed risk factors for poor adherence
- Physician (rather than nurse)
- Nursing assistant (rather than nurse)
- Male gender
- Working in intensive care unit
- Working during the week (rather than weekend)
- Wearing gowns/glove
- Automated sink
- Activities with high risk of cross contamination
- High number of hand hygiene opportunities per hour of patient care
Self-reported factors for poor adherence
- Handwashing agents cause irritation and dryness
- Sinks inconveniently located
- Lack of soap and paper towels
- Too busy
- Understaffing or overcrowding
- Patient needs take priority
- Hand hygiene interferes with patient/healthcare worker relationship
- Low risk of acquiring infection from patient
- Believing that wearing gloves means hand hygiene is unnecessary
- Lack of knowledge
- Forgetfulness
- Lack of role model
- Skepticism about need for hand hygiene
- Disagreement with recommendations
- Lack of scientific data to back up need for hand hygiene
For healthcare workers, adherence to hand hygiene guidelines protects both the patient and the worker. Hand hygiene should be done when:
- You first come on duty
- Before you touch your first patient or clean equipment
- Before and after every patient contact, including after touching intact skin
- Before any clean or invasive procedure
- Before putting on sterile gloves
- Before contact with any portal of entry, your patient’s or your own
- After contact with any body fluids, including your own
- After unprotected contact with mucous membranes or non-intact skin
- Each time you remove your gloves
- When leaving an isolation room
- When going from a dirtier to a cleaner part of the patient
- When your hands feel or look dirty
- After contact with contaminated things or environments, such as charts
- After handling used equipment or linen
- After using the bathroom
- Before and after eating
- Before going off duty
My 5 Moments for Hand Hygiene
The approach of My 5 Moments for Hand Hygiene defines the key moments when healthcare workers should perform hand hygiene. This evidence-based, field-tested, user-centered approach is designed to be easy to learn, logical, and applicable in a wide range of settings.
This approach recommends health-care workers to clean their hands:
- Before touching a patient
- Before clean/aseptic procedures
- After body fluid exposure/risk
- After touching a patient
- After touching patient surroundings (Sax et al., 2007)
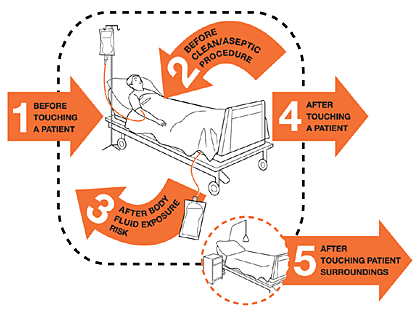
Source: Copyright © World Health Organization. Used by permission.
Alcohol Sanitizer or Soap and Water
If you can see dirt on your hands—whether from blood, body fluid, or other visible soiling—wash your hands with soap and water, which physically removes the dirt from your hands. Washing with soap and water does not kill germs.
Alcohol hand rubs do kill most germs including viruses, but they do not remove dirt and debris from your hands. If you use alcohol, choose a hand hygiene product that contains alcohol; plain alcohol should not be used because it evaporates too quickly to provide enough contact time to kill germs.
How to do hand hygiene correctly:
- If using soap, wet your hands first to minimize skin irritation.
- Use friction on all surfaces to loosen dirt and germs.
- Scrub for at least 20 seconds (Row, Row, Row Your Boat, twice).
- Use a comfortable water temperature; water hot enough to kill germs would injure your skin.
- Use alcohol hand rubs on dry skin only.
- Use one measured amount of alcohol sanitizer, and rub until hands are completely dry; do not wipe off with a paper towel.
WHO: How to Handwash with Soap and Water [1:26]
https://www.youtube.com/watch?v=3PmVJQUCm4E
For routine hand hygiene, alcohol products are preferred. Alcohol products generally are better than soap and water because:
- They kill germs more effectively.
- They leave skin in better condition.
- They are quicker and easier so are used more frequently.
How to Handwash with Alcohol-Based Formulation [1:18]
https://www.youtube.com/watch?v=ZnSjFr6J9HI&feature=emb_rel_end
After using the toilet, or when dealing with diarrhea that may be infectious, use soap and water. Both Clostridium difficile and norovirus cause diarrhea and neither is well-killed by alcohol-based hand rubs.
Because alcohol products are effective antimicrobial agents when containing more than 60% alcohol, CDC does not specify an antimicrobial soap for routine hand hygiene. Antimicrobial soaps are often more irritating to the skin, are more expensive, and tend to build up in the environment. “Plain” soap removes germs from the hands as well as an antimicrobial product.
