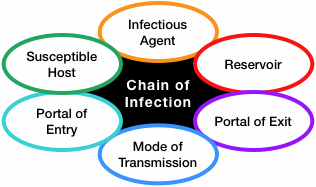
The spread of an infection within a community is described as a “chain,” several interconnected steps that describe how a pathogen moves about. Infection control and contact tracing are meant to break the chain, preventing a pathogen from spreading.
Emerging infectious diseases are those whose incidence in humans has increased in the past two decades or are a threat to increase in the near future. These diseases, which can rapidly spread across national boundaries and communities, may challenge the ability of public health systems to prevent and control the spread of the disease, especially in resource-limited countries and regions.
The spread of infection can be described as a chain with six links:
- Infectious agent (pathogen)
- Reservoir (the normal location of the pathogen)
- Portal of exit from the reservoir
- Mode of transmission
- Portal of entry into a host
- Susceptible host
Infection control measures are designed to break the links and thereby prevent a pathogen from spreading.
Infectious Agents
Infectious agents (pathogens) include not only bacteria but also viruses, fungi, and parasites. The virulence of these pathogens depends on their number, their potency, their ability to enter and survive in the body, and the susceptibility of the host. For example, the smallpox virus is particularly virulent, infecting almost all people exposed. In contrast, the tuberculosis bacillus infects only a small number of people, usually people with weakened immune function, or those who are undernourished and living in crowded conditions.
Viruses are intracellular parasites; that is, they can only reproduce inside a living cell. Some viruses, such as HIV and hepatitis B and C, have the ability to enter and survive in the body for years before symptoms of disease occur. Other viruses, such as influenza and COVID-19, quickly announce their presence through characteristic symptoms.
Reservoir
A reservoir is any person, animal, arthropod, plant, soil or substance (or combination of these) in which an infectious agent normally lives and multiplies. The infectious agent depends on the reservoir for survival, where it can reproduce itself in such manner that it can be transmitted to a susceptible host.
Animate reservoirs include people, insects, birds, and other animals. Inanimate reservoirs include soil, water, food, feces, intravenous fluid, and equipment.
Portal of Exit
Portals of exit is the means by which a pathogen exits from a reservoir. For a human reservoir, the portal of exit can include blood, respiratory secretions, and anything exiting from the gastrointestinal or urinary tracts.
Once a pathogen has exited the reservoir, it needs a mode of transmission to transfer itself into a host. This is accomplished by entering the host through a receptive portal of entry. Transmission can be by direct contact, indirect contact, or through the air.
Transmission of respiratory infections such as COVID-19 is primarily via virus-laden fluid particles (i.e., droplets and aerosols) that are formed in the respiratory tract of an infected person and expelled from the mouth and nose during breathing, talking, singing, coughing, and sneezing. The competing effects of inertia, gravity, and evaporation determine the fate of these droplets. Large droplets settle faster than they evaporate and contaminate surrounding surfaces. Smaller droplets evaporate faster than they settle, forming droplet nuclei that can stay airborne for hours (becoming aerosolized) and may be transported over long distances (Mittal et al., 2020, July 10).
Human-to-human transmission of COVID-19 occurs primarily via three routes: (1) large particles that are expelled with sufficient momentum so as to directly impact the recipients’ mouth, nose, or conjunctiva; (2) physical contact with droplets deposited on a surface and subsequent transfer to the recipient’s respiratory mucosa; and (3) inhalation of aerosolized droplet nuclei delivered by ambient air currents. The first two routes associated with large droplets are referred to as the “droplet” and “contact” routes of transmission, whereas the third is referred to as “airborne” transmission (Mittal et al., 2020, July 10).

This photograph captures a sneeze in progress, revealing the plume of salivary droplets as they are expelled in a large cone-shaped array from this man’s open mouth, thereby dramatically illustrating the reason for covering your mouth when coughing or sneezing, in order to protect others from germ exposure. Source: James Gathany, CDC PHIL, 2009.
Airborne (Aerosol) Transmission
The principal mode by which people are infected with SARS-CoV-2 is through exposure to respiratory fluids carrying infectious virus. Exposure occurs in three principal ways: (1) inhalation of very fine respiratory droplets and aerosol particles, (2) deposition of respiratory droplets and particles on exposed mucous membranes in the mouth, nose, or eye by direct splashes and sprays, and (3) touching mucous membranes with hands that have been soiled either directly by virus-containing respiratory fluids or indirectly by touching surfaces with virus on them (CDC, 2021, May 7).
People release respiratory fluids during exhalation (e.g., quiet breathing, speaking, singing, exercise, coughing, sneezing) in the form of droplets across a spectrum of sizes. These droplets carry virus and transmit infection.
- The largest droplets settle out of the air rapidly, within seconds to minutes.
- The smallest very fine droplets, and aerosol particles formed when these fine droplets rapidly dry, are small enough that they can remain suspended in the air for minutes to hours. (CDC, 2021, May 7).
Aerosols are small particles (≤5 μm) that can rapidly evaporate in the air, leaving behind droplet nuclei that are small enough and light enough to remain suspended in the air for hours (Klompas et al., 2020). Airborne transmission can occur when the residue of evaporated droplets from an infected person remain in the air long enough to be transmitted to the respiratory tract of a susceptible host.
Once infectious droplets and particles are exhaled, they move outward from the source. The risk for infection decreases with increasing distance from the source and increasing time after exhalation. Two principal processes determine the amount of virus to which a person is exposed in the air or by touching a surface contaminated by virus (CDC, 2021, May 7):
- Decreasing concentration of virus in the air as larger and heavier respiratory droplets containing virus fall to the ground or other surfaces under the force of gravity and the very fine droplets and aerosol particles that remain in the airstream progressively mix with, and become diluted within, the growing volume and streams of air they encounter. This mixing is not necessarily uniform and can be influenced by thermal layering and initial jetting of exhalations.
- Progressive loss of viral viability and infectiousness over time influenced by environmental factors such as temperature, humidity, and ultraviolet radiation (e.g., sunlight).
Transmission of SARS-CoV-2 from inhalation of virus in the air farther than six feet from an infectious source can occur.
Airborne transmission of SARS-CoV-2 is known to occur during aerosol-generating medical procedures. The scientific community has been actively discussing and evaluating whether SARS-CoV-2 may also spread through aerosols in the absence of aerosol-generating procedures, particularly in indoor settings with poor ventilation (WHO, 2020, Jun 9).
Comparing airborne (aerosol) transmission to droplet transmission is an important issue because, if COVID-19 is easily transmitted via airborne particles, then distancing, facemasks, and shields may not be enough to protect someone from exposure to the virus.
Investigators have demonstrated that speaking and coughing produce a mixture of both droplets and aerosols in a range of sizes, that these secretions can travel together for up to 27 feet, that it is feasible for SARS-CoV-2 to remain suspended in the air and viable for hours, that SARS-CoV-2 RNA can be recovered from air samples in hospitals, and that poor ventilation prolongs the amount of time that aerosols remain airborne (Klompas et al., 2020).
During the initial isolation, of thirteen individuals from the Diamond Princess cruise ship who had COVID-19, at the University of Nebraska Medical Center, researchers collected air and surface samples to examine viral shedding from isolated individuals. They detected viral contamination among all samples, supporting the use of airborne isolation precautions when caring for COVID-19 patients (Santarpia, et al., 2020, July 29).
The presence of contamination on personal items was expected, particularly those items that are routinely handled by individuals in isolation, such as cell phones and remote controls, as well as medical equipment that is in near-constant contact with the patient. The observation of viral replication in cell culture for some of the samples confirms the potentially infectious nature of the recovered virus (Santarpia, et al., 2020, July 29).
Researchers noted variability in the degree of environmental contamination from room to room and day to day. Patients with higher acuity of illness or levels of care may be associated with increased levels of environmental contamination. However, there was a lack of a strong relationship between environmental contamination and body temperature, reaffirming the fact that shedding of viral RNA is not necessarily linked to clinical signs of illness (Santarpia, et al., 2020, July 29).
The more acute patients were generally less mobile, and distribution of positive samples suggested a strong influence of airflow. Personal and high-touch items were not universally positive, yet viral RNA was detected in 100% of samples from the floor under the bed and all but one window ledge (which were not used by the patient) (Santarpia, et al., 2020, July 29).
Data from the UNMC study indicated significant environmental contamination in rooms where patients infected with SARS-CoV-2 were housed and cared for, regardless of the degree of symptoms or acuity of illness. Contamination existed in all types of samples: high- and low-volume air samples, as well as surface samples including personal items, room surfaces, and toilets. Samples of patient toilets that tested positive for viral RNA are consistent with other reports of viral shedding in stool (Santarpia, et al., 2020, July 29).
The transport of droplet nuclei over larger distances is primarily driven by ambient air flows, and indoor environments such as homes, offices, malls, aircraft, and public transport vehicles pose a particular challenge for disease transmission. The importance of ventilation in controlling airborne transmission of infections is well known. Indoor spaces can have extremely complex flows, due to ventilation systems and other factors that influence them (Mittal et al., 2020, July 10).
Diagram Showing Airborne Transmission
Engineering level controls to reduce the environmental risks for airborne transmission. Source: Environmental International Volume 142. CC BY-NC-ND 4.0.
Indirect Contact
Indirect contact includes both vehicle-borne and vector borne contact. A vehicle is an inanimate go-between, an intermediary between the portal of exit from the reservoir and the portal of entry to the host. Inanimate objects such as cooking or eating utensils, handkerchiefs and tissues, soiled laundry, doorknobs and handles, and surgical instruments and dressings are common vehicles that can transmit infection. Blood, serum, plasma, water, food, and milk also serve as vehicles. For example, food can be contaminated by E.coli if food handlers do not practice appropriate handwashing techniques after using the bathroom. If the food is eaten by a susceptible host, such as a young child or a person with HIV/AIDS, the resulting infection can be life-threatening.
Vector-borne contact is transmission by an animate intermediary, an animal, insect, or parasite that transports the pathogen from reservoir to host. Transmission takes place when the vector injects salivary fluid by biting the host, or deposits feces or eggs in a break in the skin. Mosquitoes are vectors for malaria and West Nile virus. Rodents can be vectors for hantavirus.
Portal of Entry
Infectious agents get into the body through various portals of entry, including the mucous membranes, non-intact skin, and the respiratory, gastrointestinal, and genitourinary tracts. Pathogens often enter the body of the host through the same route they exited the reservoir, e.g., airborne pathogens from one person’s sneeze can enter through the nose of another person.
Susceptible Host
The final link in the chain of infection is a susceptible host, someone at risk of infection. Infection does not occur automatically when the pathogen enters the body of a person whose immune system is functioning normally. When a virulent pathogen enters an immune-compromised person, however, infection generally follows.
Whether exposure to a pathogen results in infection depends on several factors related to the person exposed (the host), the pathogen (the agent), and the environment. Host factors that influence the outcome of an exposure include the presence or absence of natural barriers, the functional state of the immune system, and the presence or absence of an invasive device.
How COVID-19 Spreads
From the start of the COVID-19 epidemic, it was known that the virus spreads via respiratory droplets (infectious agent). In a CDC telebriefing on February 14, CDC’s Messonnier said, “Based on what is now known about COVID-19, we believe this virus spreads mainly from person (reservoir) to person among close contacts (defined as about six feet) through respiratory droplets produced when an infected person coughs or sneezes.” This is similar to the way influenza and other respiratory pathogens spread. These droplets can land in the mouths or noses of people (susceptible host) who are nearby or possibly be inhaled into the lungs (portal of entry).
What was not initially known was that asymptomatic people can act as a reservoir for the virus, thus infecting others. Those who do develop symptoms appear to be “shedding significant virus in their oropharyngeal compartment” up to 48 hours before developing symptoms. “This helps explain how rapidly this virus continues to spread across the country, because we have asymptomatic transmitters and we have individuals who are transmitting 48 hours before they become symptomatic,” said CDC Director Robert Redfield.
A July 9, 2020 scientific brief from the World Health Organization titled Transmission of SARS-CoV-2: Implications for Infection Prevention Precautions, indicates that infectious particles much smaller than droplets can become airborne and remain suspended in the air for long periods of time. These so-called aerosol particles appear to be capable of infecting people who inhale them. WHO reports that outbreaks related to activities in crowded, enclosed spaces “suggest the possibility of aerosol transmission combined with droplet transmission” during activities such as choir practice, eating in restaurants, and exercising in gyms.
Viability on Surfaces
The principal mode by which people are infected with SARS-CoV-2 (the virus that causes COVID-19) is through exposure to respiratory droplets carrying infectious virus. It is possible for people to be infected through contact with contaminated surfaces or objects (fomites), but the risk is generally considered to be low (CDC, 2021, April 5).
