
Source: CDC.
Tests are used in community, outpatient, and hospital-based surveillance systems to identify cases of SARS-CoV-2 infection. Data from tests can identify areas of ongoing circulation, determine trends, provide insight into the impact of the disease over time and inform disease forecasts. Clinical criteria for considering testing have been developed based on what is known about COVID-19 and are subject to change as additional information becomes available (CDC, 2020, July 2).
As noted earlier, two kinds of tests are available for COVID-19:
- Diagnostic viral test tells you if you have a current infection and need to take steps to quarantine or isolate yourself. There are two main viral tests:
- Molecular (RT-PCR)
- Antigen tests
- Antibody test tells you if you had a previous infection. It looks for antibodies made by the immune system in response to the viral threat.
Viral Diagnostic Tests
Viral tests check samples (such as a nasal swab) from the respiratory system to determine whether an infection with SARS-CoV-2 is present. Viral tests are used to diagnose acute infection (CDC, 2020 July 2). Ideally, diagnostic testing would be conducted for all patients with a syndrome consistent with COVID-19, people with known high-risk exposures, and people likely to be at repeated risk of exposure, such as healthcare workers and first responders (NAIAD-RML, 2020, July 30).
Gathering a Respiratory Specimin
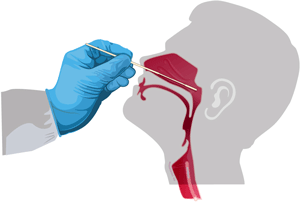
Nasopharyngeal swab. Source: CDC.
Viral tests can be point-of-care tests, meaning results may be available at the testing site in less than an hour. Other viral tests must be sent to a lab for analysis, a process that takes 1 to 2 days (or longer) once received by the lab.
CDC recommends that nasopharynx samples be used to detect SARS-CoV-2. Nasal swabs or oropharyngeal swabs may be acceptable alternatives. Lower–respiratory tract samples have a higher yield than upper-tract samples, but often they are not obtained because of concerns about aerosolization of virus during sample collection procedures (NAIAD-RML, 2020, July 30).
While initial diagnostic tests for SARS-CoV-2 infection have relied on reverse transcriptase polymerase chain reaction platforms, more recent tests have included a variety of additional platforms. More than twenty diagnostic tests for SARS-CoV-2 infection have received Emergency Use Authorization by the Food and Drug Administration. Formal comparisons of these tests are in progress (NAIAD-RML, 2020, July 30).
Molecular diagnostic and antigen tests can yield false-negative results. In people with a high likelihood of infection based on exposure history and/or clinical presentation, a single negative test result does not completely exclude SARS-CoV-2 infection and repeat testing should be considered. When a person who is strongly suspected to have SARS-CoV-2 infection has a negative result on an initial antigen test, repeat testing using a molecular diagnostic test may be warranted (NIH, 2020, Jun 11).
COVID-19 Diagnostics: Performing a Nasopharyngeal and Oropharyngeal Swab [3:54]
https://www.youtube.com/watch?v=syXd7kgLSN8&feature=emb_rel_end
Molecular Diagnostic Tests
Molecular diagnostic testing combines laboratory testing with the precision of molecular biology to investigate the human, viral, and microbial genomes, their genes, and the products they encode. Molecular diagnostic tests are increasingly being used, and have supplanted numerous conventional tests, in many areas of laboratory medicine including oncology, infectious diseases, clinical chemistry, and clinical genetics (CDC, 2019, Nov 5).
SARS Test Kit
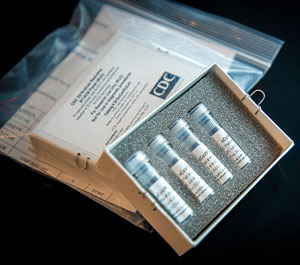
CDC’s laboratory test kit for the SARS-CoV-2 virus. Source: CDC, 2020, July 15.
Molecular tests detect a virus’s genetic material. They are used to diagnose COVID-19 (or active coronavirus infection) using a sample from the patient’s nose or throat. The CDC 2019 Novel Coronavirus Real-Time Reverse Transcriptase (RT)–PCR Diagnostic Panel detects the SARS-CoV-2 virus in upper -and lower-respiratory specimens. It is designed to be used with an existing RT-PCR testing instrument commonly used to test for seasonal influenza virus (CDC, 2020, July 15.)
A positive test result for COVID-19 indicates that RNA from SARS-CoV-2 was detected, and the patient is presumptively infected with the virus and presumed to be contagious. Laboratory test results should always be considered in the context of clinical observations and epidemiologic data in making final diagnosis and patient management decisions. Patient management decisions should be made with a healthcare provider and follow current CDC guidelines (CDC, 2020, June 12).
A negative test result for this test means that SARS-CoV-2 RNA was not present in the specimen above the limit of detection. However, a negative result does not rule out COVID-19 and should not be used as the sole basis for treatment or patient management decisions. A negative result does not exclude the possibility of COVID-19 (CDC, 2020, June 12).
When diagnostic testing is negative, the possibility of a false negative result should be considered in the context of a patient’s recent exposures and the presence of clinical signs and symptoms consistent with COVID-19. The possibility of a false negative result should especially be considered if the patient’s recent exposures or clinical presentation indicate that COVID19 is likely, and diagnostic tests for other causes of illness (e.g., other respiratory illness) are negative. If COVID-19 is still suspected based on exposure history together with other clinical findings, re-testing should be considered by healthcare providers in consultation with public health authorities (CDC, 2020, June 12).
Molecular tests are not 100 percent accurate due to these factors:
- The swab might not collect the virus from a person’s nose or throat.
- The swab or mucus sample may be accidentally contaminated by the virus during collection or analysis.
- The nasal or throat swab may not be kept at the correct temperature before it can be analyzed.
- The chemicals used to extract the virus genetic material and make copies of the virus DNA may not work correctly. (FDA, 2020, July 16)
Antigen Tests
Antigens are molecules on the surface of viruses that trigger an immune response. Antigens have specific surface features that are recognized by the immune system. The SARS-CoV-2 virus has several surface antigens, one of which is the “spike protein” visible on the surface of the virus. When a person is infected with COVID-19, the immune system begins to produce antibodies, which attack the surface antigens.
Antigen tests usually provide results faster than molecular tests, but antigen tests have a higher chance of missing an active infection. If an antigen test shows a negative result, indicating that you do not have an active coronavirus infection, your healthcare provider may order a molecular test to confirm the result (FDA, 2020, July 16).
Priorities for Diagnostic Testing
CDC has established a priority system for diagnostic testing for SARS-CoV-2 infection based on the availability of tests; the CDC testing guidance is updated periodically.
High priority
- Hospitalized patients with symptoms
- Healthcare facility workers, workers in congregate living settings, and first responders with symptoms
- Residents in long-term care facilities or other congregate living settings, including prisons and shelters, with symptoms
Priority
- Persons with symptoms of potential COVID-19 infection, including fever, cough, shortness of breath, chills, muscle pain, new loss of taste or smell, vomiting or diarrhea, and/or sore throat
- Persons without symptoms who are prioritized by health departments or clinicians, for any reason, including but not limited to public health monitoring, sentinel surveillance, or screening of other asymptomatic individuals according to state and local plans (NIH, 2020, Jun 11)
Antibody Tests
Antibody tests, a type of serologic test, detect antibodies present in the blood when the body is responding to or has responded to a specific infection. Antibody tests detect the body’s immune response to the infection caused by the virus rather than detecting the virus itself. Antibody tests should not be used to diagnose current SARS-CoV-2 infection (FDA, 2020, May 4).
Antibody Test
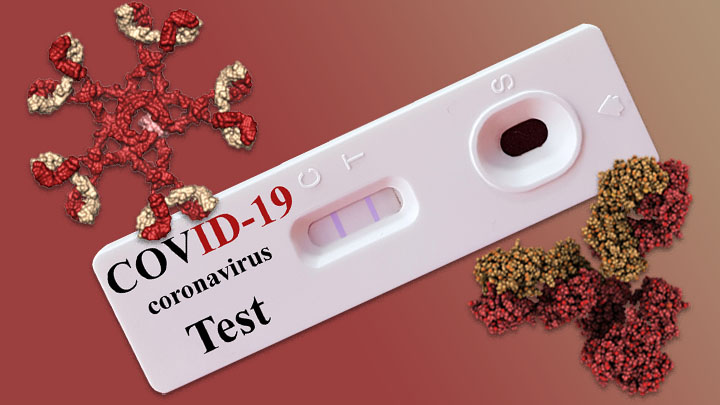
An image showing two antibodies (red/gold) with a white COVID-19 test cartridge in the middle. Source: NIH Director’s Blog. Public domain.
Antibody tests can be used to determine if individual patients may have been exposed to and infected with a virus, and also can be used to understand how many people in a population have antibodies (then they are known as surveillance tests, or sero-surveys) (FDA, 2020, May 4). Antibodies can take several days or weeks to develop after an infection and may stay in the blood for several weeks or more after recovery.
People who have developed antibodies against SARS-CoV-2 may qualify to donate blood that can be used to manufacture convalescent plasma, an investigational product for use with those who are seriously ill from COVID-19 (FDA, 2020, May 4). In general, a positive antibody test is presumed to mean a person has been infected with SARS-CoV-2 at some point in the past. It does not mean they are currently infected. Antibodies start developing within 1 to 3 weeks after infection. It is not known whether someone will be immune and protected from reinfection if they have antibodies to the virus (CDC, 2020, May 28).
Here is a summary of CDC advice for healthcare providers, laboratory professionals, and public health professionals using antibody tests for SARS-CoV-2, the virus that causes COVID-19.
- Choose antibody tests that have Emergency Use Authorization (EUA) from the U.S. Food and Drug Administration.
- Do not use antibody tests to determine a person’s immune status until evidence confirms that antibodies provide protection, how much antibody is protective, and how long protection lasts.
- Antibody test results should not be used to diagnose someone with an active infection.
- Antibody tests can support the clinical assessment of COVID-19 illness for people who are being tested 9 to 14 days after illness onset, in addition to recommended virus detection methods such as PCR. This will maximize sensitivity because the sensitivity of nucleic acid detection is decreasing and serologic testing is increasing during this time period.
- Antibody testing can help establish a clinical picture when patients have late complications of COVID-19 illness, such as multisystem inflammatory syndrome in children. (CDC, 2020, May 28)
Caveat
Antibody test results should not be used to determine if someone can return to work. Antibody test results should not be used to group people together in settings such as schools, dormitories, and correctional facilities.
Source: CDC, 2020, May 28.
Antibody tests are not authorized by FDA for such diagnostic purposes. In certain situations, serologic assays may be used to support clinical assessment of persons who present late in their illnesses when used in conjunction with viral detection tests. In addition, if a person is suspected to have post-infectious syndrome caused by SARS-CoV-2, serologic assays may be used (CDC, 2020, July 2)
Serologic assays for SARS-CoV-2 can play an important role in understanding the transmission dynamic of the virus in the general population and identifying groups at higher risk for infection. Unlike viral direct detection methods, such as nucleic acid amplification or antigen detection tests that can detect acutely infected persons, antibody tests help determine whether the individual being tested was previously infected—even if that person never showed symptoms (CDC, 2020, July 2).
Different Types of Coronavirus Tests | |||
|---|---|---|---|
| Molecular test | Antigen test | Antibody test |
Also known as… | Diagnostic test, viral test, molecular test, nucleic acid amplification tests (NAAT), RT-PCR tests, LAMP test | Rapid diagnostic test (Some molecular tests are also rapid tests.) | Serological test, serology, blood test, serology test |
How sample taken… | Nasal or throat swab (most tests) Saliva (a few tests) | Nasal or throat swab | Finger stick or blood draw |
How long for results… | Same day (some locations) or up to a week | One hour or less | Same day (many locations) or 1-3 days |
Is another test needed… | This test is typically highly accurate and usually does not need to be repeated. | Positive results are usually highly accurate but negative results may need to be confirmed with a molecular test | Sometimes a second antibody test is needed for accurate results. |
What it shows… | Diagnoses active coronavirus infection | Diagnoses active coronavirus infection | Shows if you’ve been infected by coronavirus in the past |
What it can’t do… | Show if you ever had COVID-19 or were infected with the coronavirus in the past | Definitively rule out active coronavirus infection. Antigen tests are more likely to miss an active coronavirus infection compared to molecular tests. Your health care provider may order a molecular test if your antigen test shows a negative result, but you have symptoms of COVID-19. | Diagnose active coronavirus infection at the time of the test or show that you do not have COVID-19 |
When to Test
[Material in this section is from the CDC: Evaluating and Testing Persons for Coronavirus Disease 2019 unless otherwise noted.]
Clinicians should use their judgment to determine if a patient has signs and symptoms compatible with COVID-19 and whether the patient should be tested. Asymptomatic infection with SARS-CoV-2 has been reported. Most patients with confirmed COVID-19 have developed fever and/or symptoms of acute respiratory illness but some people may present with other symptoms as well.
Other considerations that may guide testing are epidemiologic factors such as known exposure to an individual who has tested positive for SARS-CoV-2, and the occurrence of local community transmission or transmission within a specific setting or facility. Clinicians are strongly encouraged to test for other causes of respiratory illness, such as influenza, in addition to testing for SARS-CoV-2. Another population in which to prioritize testing of minimally symptomatic and even asymptomatic persons are long-term care facility residents, especially in facilities where one or more other residents have been diagnosed with symptomatic or asymptomatic COVID-19.
SARS-CoV-2 can cause asymptomatic, pre-symptomatic, and minimally symptomatic infections, leading to viral shedding that may result in transmission to others who are particularly vulnerable to severe disease and death. Even mild signs and symptoms should be evaluated among potentially exposed healthcare personnel, due to their extensive and close contact with vulnerable patients in healthcare settings.
New Testing Technologies
The National Institutes of Health is investing $248.7 million in new technologies to address challenges associated with COVID-19 testing. NIH’s Rapid Acceleration of Diagnostics (RADx) initiative has awarded contracts to seven biomedical diagnostic companies to support a range of new lab-based and point-of-care tests that could significantly increase the number, type, and availability of tests by millions per week as early as September 2020. With national demand estimated to be millions more tests per day above current levels, these technologies are expected to make a significant contribution to expanding the nation’s testing capacity (NIH, 2020, July 31).
The U.S. Food and Drug Administration granted emergency use authorization (EUA) on December 15, 2020 for an innovative COVID-19 viral antigen test developed with support from the RADx Initiative. Ellume USA designed the test for use at home without a prescription. This is the first EUA awarded for an at-home COVID test that can be purchased over the counter. Ellume developed the test with a $30 million contract and technical support from the RADx Tech program, managed by the National Institute of Biomedical Imaging and Bioengineering, part of NIH (NIH, 2020, December 15).
The test is performed using a mid-turbinate nasal swab designed for comfortable self-sampling. The sample is inserted into a single-use cartridge that returns results in 15 minutes. The at-home test analyzer connects to the user’s smartphone through Bluetooth and pairs with a downloadable app that provides step-by-step instructions and displays results (NIH, 2020, December 15).
Users can share real-time results from the test, selling for approximately $30, with healthcare professionals, employers, and schools for efficient COVID-19 tracking. Ellume plans to scale-up manufacturing to deliver millions of home tests per month in 2021 (NIH, 2020, December 15).
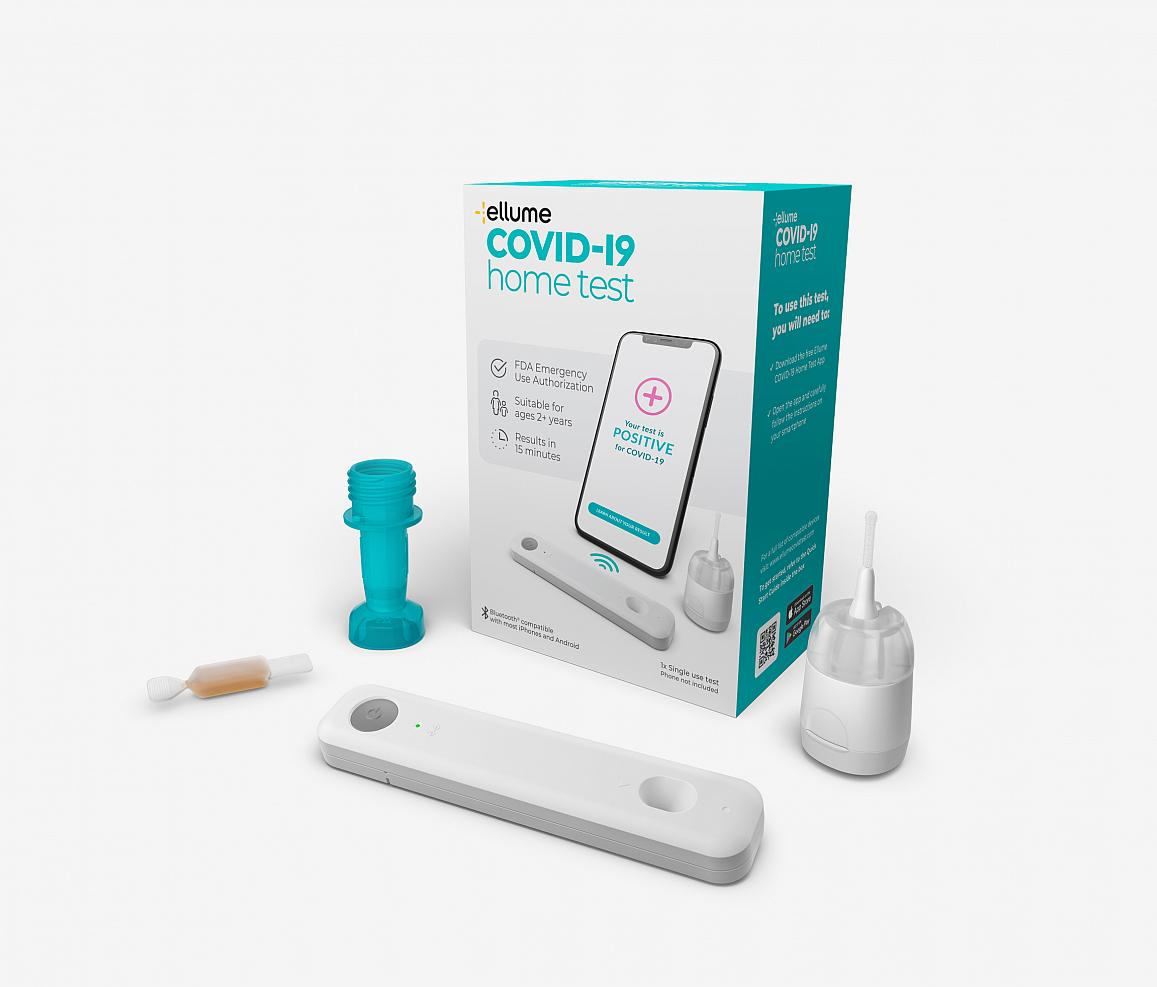
Source: NIH
Here are some other types of diagnostic tests available:
- Rapid, point-of-care diagnostic tests use a mucus sample from the nose or throat but can be analyzed at the doctor’s office or clinic where the sample is collected, and results may be available in minutes. These may be molecular or antigen tests.
- At-home collection tests, available only by prescription from a doctor, allow the patient to collect the sample at home and send it directly to the lab for analysis.
- Saliva tests allow a patient to spit into a tube rather than get their nose or throat swabbed. Saliva tests may be more comfortable for some people and may be safer for healthcare workers who can be farther away during the sample collection. (FDA, 2020, July 16)
Point-of-Care Testing

Quidel’s Sofia 2 point-of-care instrument delivers test results for COVID-19 in 15 minutes. Source: NIH.
Visual Read Test to Detect Virus
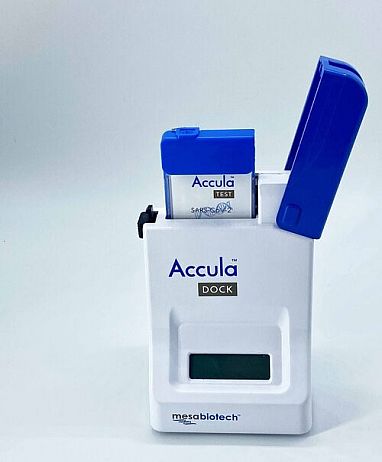
Mesa Biotech’s Accula System is a visually read test using RT-PCR technology to detect SARS-CoV-2 at the point of care and provides lab-quality results in about 30 minutes. Source: NIH.
